QUESTION IMAGE
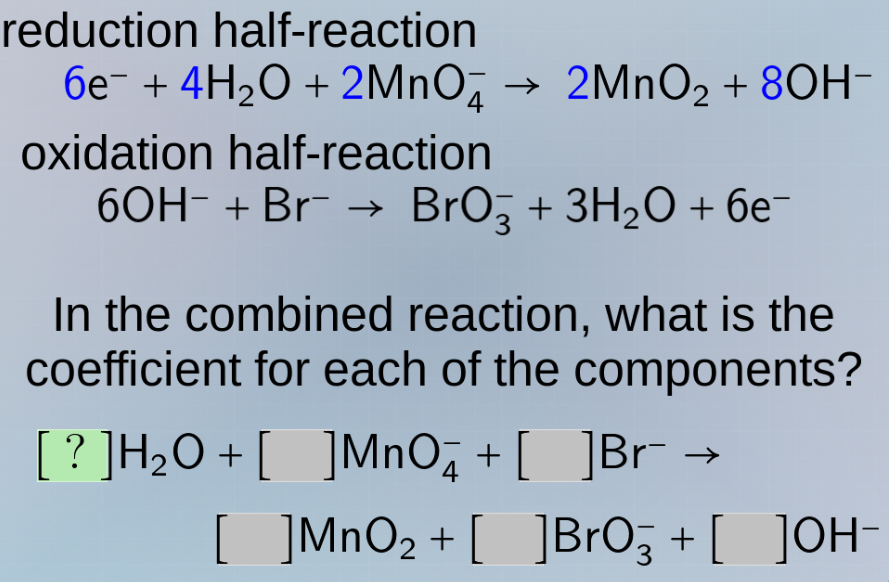
Question
reduction half - reaction
$6\ce{e-} + 4\ce{h2o} + 2\ce{mno4-} \
ightarrow 2\ce{mno2} + 8\ce{oh-}$
oxidation half - reaction
$6\ce{oh-} + \ce{br-} \
ightarrow \ce{bro3-} + 3\ce{h2o} + 6\ce{e-}$
in the combined reaction, what is the coefficient for each of the components?
$?\ce{h2o} + \quad\ce{mno4-} + \quad\ce{br-} \
ightarrow
\quad\ce{mno2} + \quad\ce{bro3-} + \quad\ce{oh-}$
Step1: Add the two half - reactions
The reduction half - reaction is \(6e^- + 4H_2O+2MnO_4^-
ightarrow2MnO_2 + 8OH^-\)
The oxidation half - reaction is \(6OH^-+Br^-
ightarrow BrO_3^-+3H_2O + 6e^-\)
When we add these two half - reactions, the electrons (\(6e^-\)) will cancel out.
For the \(H_2O\) molecules: In the reduction half - reaction, we have \(4H_2O\), and in the oxidation half - reaction, we have \(3H_2O\) on the product side. So the net number of \(H_2O\) molecules on the reactant side is \(4H_2O- 3H_2O=1H_2O\) (because we are adding the two reactions, the \(3H_2O\) on the product side of the oxidation reaction is equivalent to subtracting \(3H_2O\) from the reactant side when combining).
For \(MnO_4^-\): We have \(2MnO_4^-\) from the reduction half - reaction.
For \(Br^-\): We have \(1Br^-\) from the oxidation half - reaction.
For \(MnO_2\): We have \(2MnO_2\) from the reduction half - reaction.
For \(BrO_3^-\): We have \(1BrO_3^-\) from the oxidation half - reaction.
For \(OH^-\): In the reduction half - reaction, we have \(8OH^-\) on the product side, and in the oxidation half - reaction, we have \(6OH^-\) on the reactant side. So the net number of \(OH^-\) on the product side is \(8OH^--6OH^- = 2OH^-\)
Step2: Verify the combination
After adding the two half - reactions:
\(H_2O+2MnO_4^-+Br^-
ightarrow2MnO_2 + BrO_3^-+2OH^-\)
Let's check the atom counts:
- Mn: Left side (from \(MnO_4^-\)): 2, Right side (from \(MnO_2\)): 2
- Br: Left side (from \(Br^-\)): 1, Right side (from \(BrO_3^-\)): 1
- O: Left side: \(1\times1\) (from \(H_2O\)) \(+2\times4\) (from \(MnO_4^-\)) \(=1 + 8=9\); Right side: \(2\times2\) (from \(MnO_2\)) \(+3\) (from \(BrO_3^-\)) \(+2\times1\) (from \(OH^-\)) \(=4 + 3+2 = 9\)
- H: Left side: \(1\times2\) (from \(H_2O\)) \(= 2\); Right side: \(2\times1\) (from \(OH^-\)) \(= 2\)
- Charge: Left side: \(2\times(- 1)\) (from \(MnO_4^-\)) \(+(-1)\) (from \(Br^-\)) \(=-2-1=-3\); Right side: \(2\times(-1)\) (from \(OH^-\)) \(+(-1)\) (from \(BrO_3^-\)) \(=-2 - 1=-3\) (charge is balanced)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
1 \(H_2O\) + 2 \(MnO_4^-\) + 1 \(Br^-\) \(
ightarrow\) 2 \(MnO_2\) + 1 \(BrO_3^-\) + 2 \(OH^-\)
So the coefficients are: \(H_2O\): 1, \(MnO_4^-\): 2, \(Br^-\): 1, \(MnO_2\): 2, \(BrO_3^-\): 1, \(OH^-\): 2