QUESTION IMAGE
Question
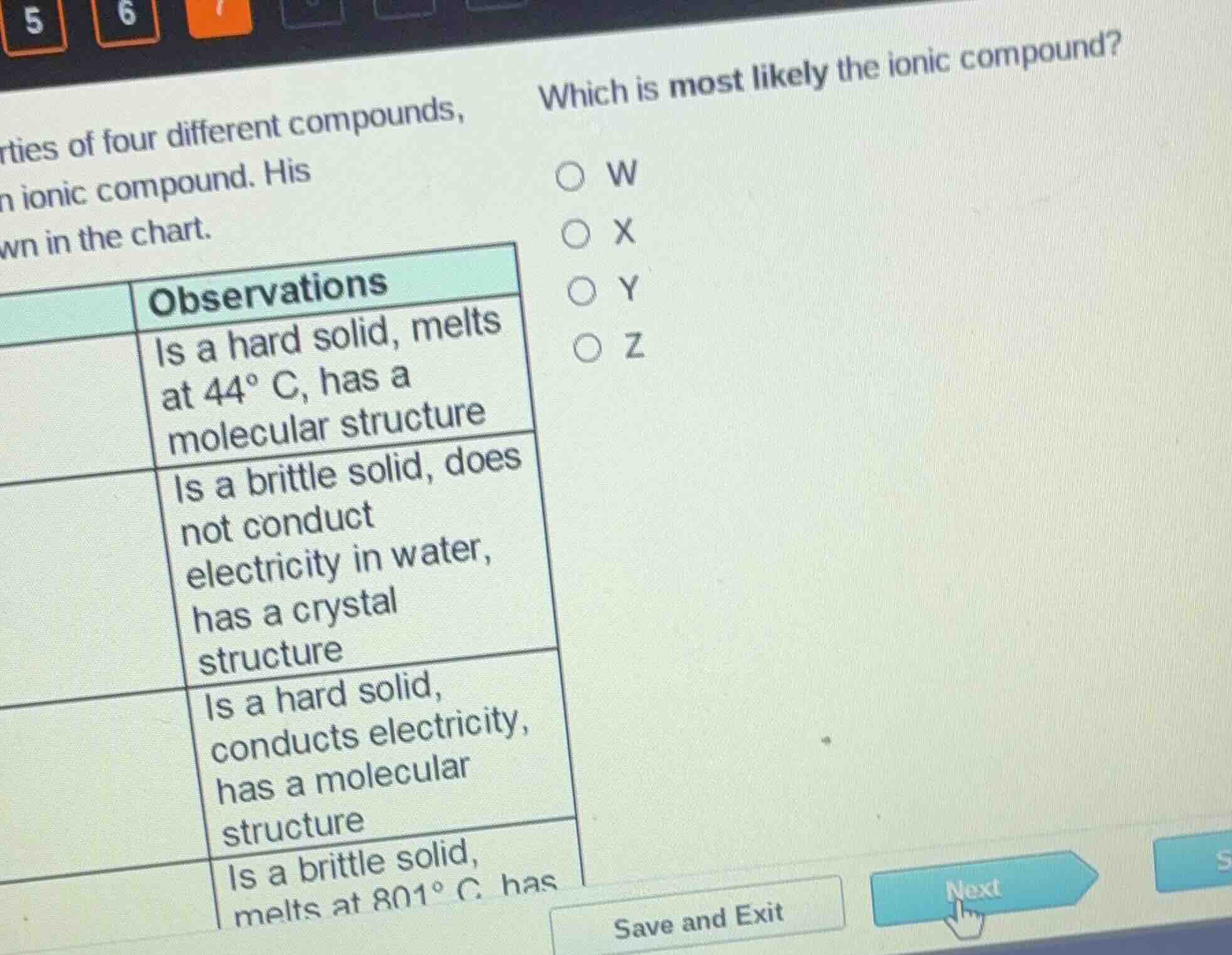
rties of four different compounds,
ionic compound. his
wn in the chart.
which is most likely the ionic compound?
observations
is a hard solid, melts
at 44° c, has a
molecular structure
is a brittle solid, does
not conduct
electricity in water,
has a crystal
structure
is a hard solid,
conducts electricity,
has a molecular
structure
is a brittle solid,
melts at 801° c, has
w
x
y
z
Step1: Recall ionic compound properties
Ionic compounds are brittle solids, have high melting points (usually >300°C), form crystal lattices, and conduct electricity when dissolved in water (due to ions).
Step2: Analyze each compound
- Compound W: Hard solid, melts at 44°C, molecular structure. Low melting point and molecular structure → not ionic.
- Compound X: Brittle solid, doesn't conduct in water, crystal structure. Ionic compounds conduct in water (ions), so this is inconsistent → not ionic.
- Compound Y: Hard solid, conducts electricity, molecular structure. Molecular compounds don't conduct (no ions) → not ionic.
- Compound Z: Brittle solid, melts at 801°C (high), and (implied) likely has ionic bonding (crystal structure, high melting, brittle). Matches ionic properties.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Z