QUESTION IMAGE
Question
section 4: full balancing challenge (show mastery)
this is where everything comes together.
directions:
balance each equation completely.
✓ coefficients only
✓ smallest whole numbers
✓ check your work
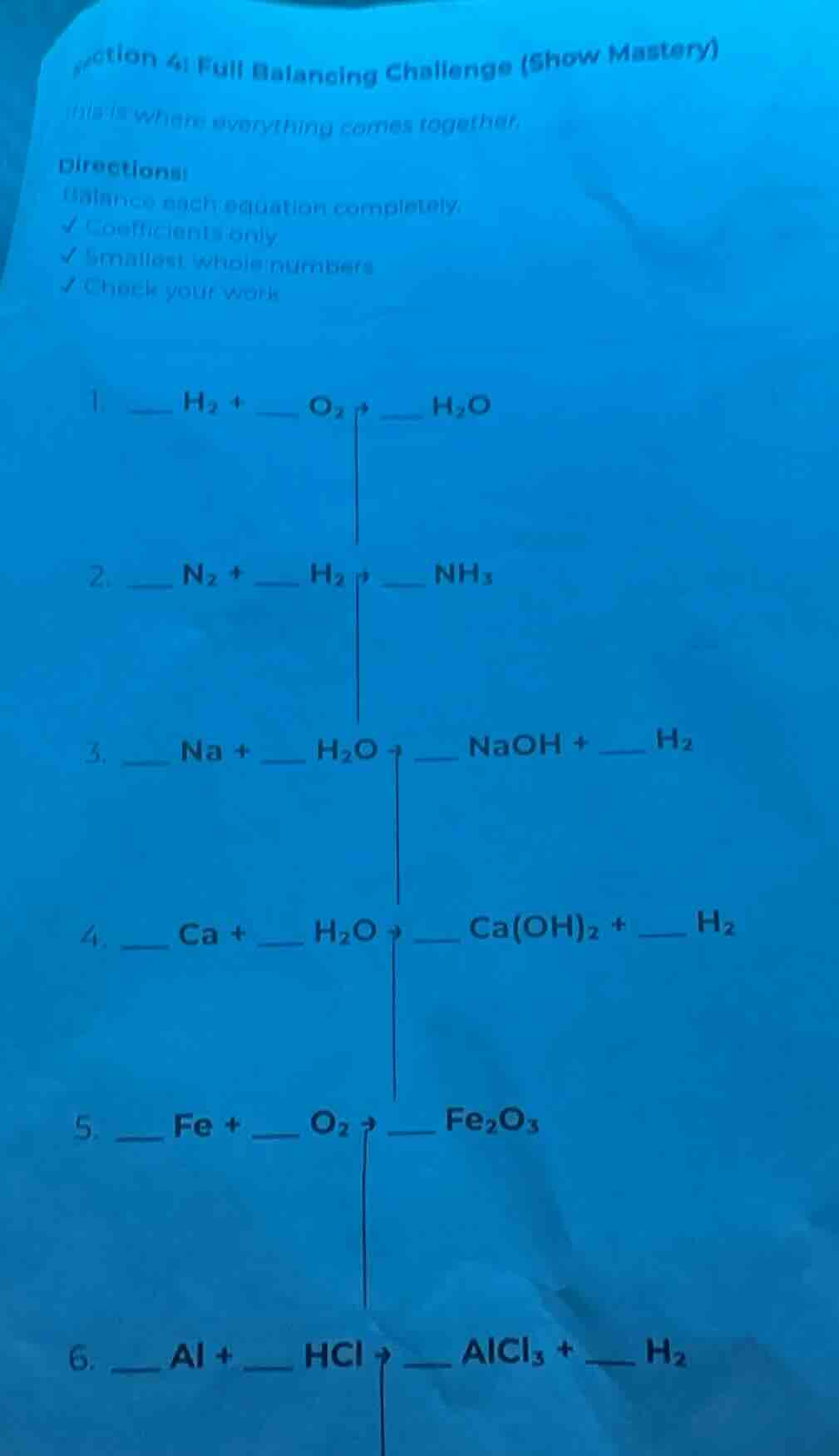
1 _ h₂ + _ o₂ → ___ h₂o
- _ n₂ + _ h₂ → ___ nh₃
- _ na + _ h₂o → _ naoh + _ h₂
- _ ca + _ h₂o → _ ca(oh)₂ + _ h₂
- _ fe + _ o₂ → ___ fe₂o₃
- _ al + _ hcl → _ alcl₃ + _ h₂
Step1: Balance H₂ + O₂ → H₂O
Count O: 2 on left, 1 on right. Add 2 to H₂O: $\text{H}_2 + \text{O}_2
ightarrow 2\text{H}_2\text{O}$. Now H: 2 on left, 4 on right. Add 2 to H₂: $2\text{H}_2 + \text{O}_2
ightarrow 2\text{H}_2\text{O}$
Step2: Balance N₂ + H₂ → NH₃
Count N: 2 on left, 1 on right. Add 2 to NH₃: $\text{N}_2 + \text{H}_2
ightarrow 2\text{NH}_3$. Now H: 2 on left, 6 on right. Add 3 to H₂: $\text{N}_2 + 3\text{H}_2
ightarrow 2\text{NH}_3$
Step3: Balance Na + H₂O → NaOH + H₂
Count H: 2 on left, 3 on right. Add 2 to H₂O and NaOH: $\text{Na} + 2\text{H}_2\text{O}
ightarrow 2\text{NaOH} + \text{H}_2$. Now Na: 1 on left, 2 on right. Add 2 to Na: $2\text{Na} + 2\text{H}_2\text{O}
ightarrow 2\text{NaOH} + \text{H}_2$
Step4: Balance Ca + H₂O → Ca(OH)₂ + H₂
Count O: 2 in Ca(OH)₂, add 2 to H₂O: $\text{Ca} + 2\text{H}_2\text{O}
ightarrow \text{Ca(OH)}_2 + \text{H}_2$. Check H: 4 on left, 4 on right; Ca: 1 on both sides.
Step5: Balance Fe + O₂ → Fe₂O₃
Count O: 2 on left, 3 on right. Use least common multiple 6: add 3 to O₂, 2 to Fe₂O₃: $\text{Fe} + 3\text{O}_2
ightarrow 2\text{Fe}_2\text{O}_3$. Now Fe: 1 on left, 4 on right. Add 4 to Fe: $4\text{Fe} + 3\text{O}_2
ightarrow 2\text{Fe}_2\text{O}_3$
Step6: Balance Al + HCl → AlCl₃ + H₂
Count Cl: 3 in AlCl₃, add 3 to HCl: $\text{Al} + 3\text{HCl}
ightarrow \text{AlCl}_3 + \text{H}_2$. Now H: 3 on left, 2 on right. Use least common multiple 6: add 2 to Al, 6 to HCl, 2 to AlCl₃, 3 to H₂: $2\text{Al} + 6\text{HCl}
ightarrow 2\text{AlCl}_3 + 3\text{H}_2$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- $2\text{H}_2 + \text{O}_2
ightarrow 2\text{H}_2\text{O}$
- $\text{N}_2 + 3\text{H}_2
ightarrow 2\text{NH}_3$
- $2\text{Na} + 2\text{H}_2\text{O}
ightarrow 2\text{NaOH} + \text{H}_2$
- $\text{Ca} + 2\text{H}_2\text{O}
ightarrow \text{Ca(OH)}_2 + \text{H}_2$
- $4\text{Fe} + 3\text{O}_2
ightarrow 2\text{Fe}_2\text{O}_3$
- $2\text{Al} + 6\text{HCl}
ightarrow 2\text{AlCl}_3 + 3\text{H}_2$