QUESTION IMAGE
Question
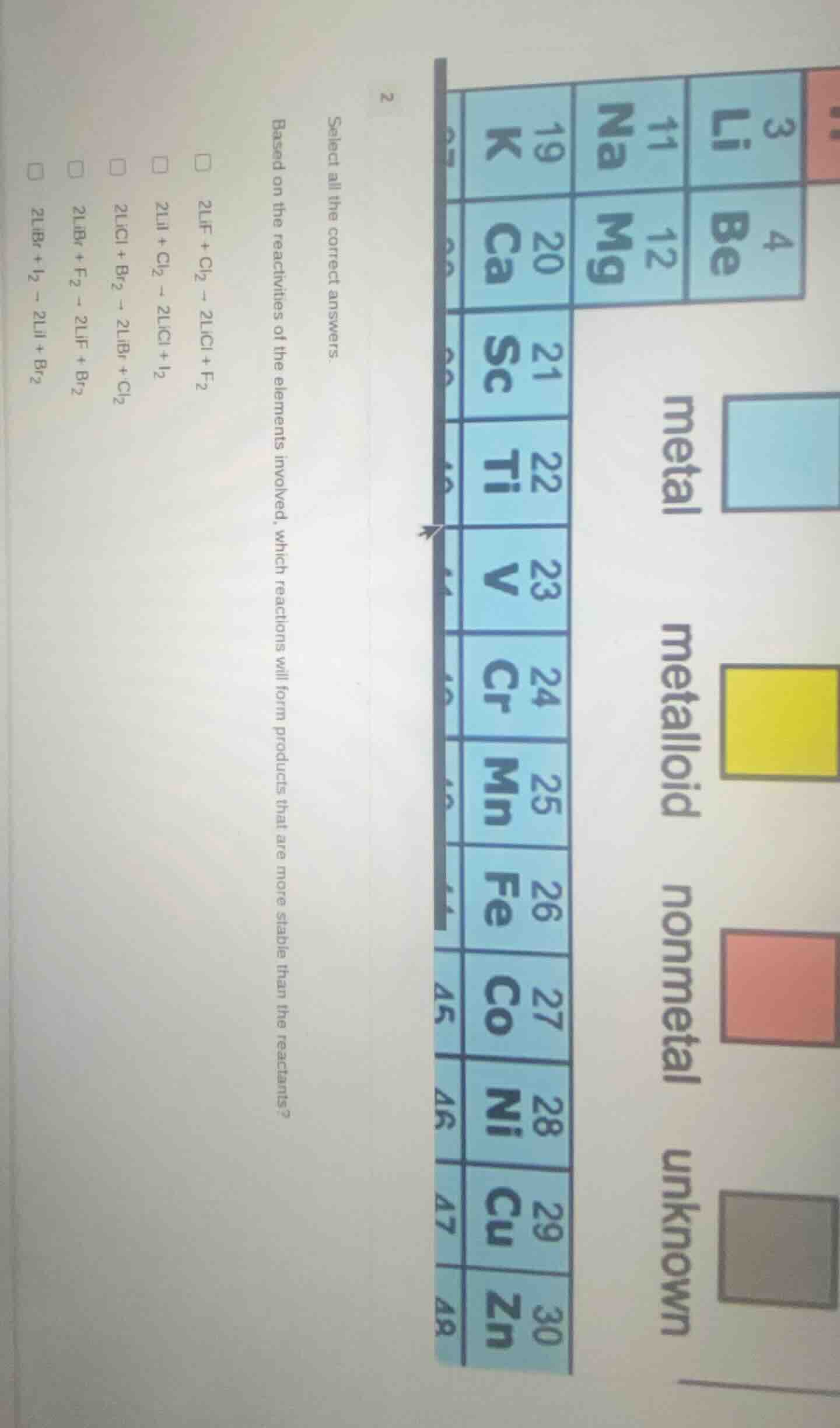
2 select all the correct answers. based on the reactivities of the elements involved, which reactions will form products that are more stable than the reactants? 2lif + cl₂ → 2licl + f₂ 2lii + cl₂ → 2licl + i₂ 2licl + br₂ → 2libr + cl₂ 2libr + f₂ → 2lif + br₂ 2libr + i₂ → 2lii + br₂
To solve this, we analyze the reactivity of halogens (F, Cl, Br, I) from the periodic table. Halogens become less reactive down the group: \( \text{F}_2 > \text{Cl}_2 > \text{Br}_2 > \text{I}_2 \). A more reactive halogen can displace a less reactive one from its salt.
Analyze each reaction:
- **\( 2\text{LiF} + \text{Cl}_2
ightarrow 2\text{LiCl} + \text{F}_2 \)**
\( \text{Cl}_2 \) is less reactive than \( \text{F}_2 \) (since \( \text{F}_2 > \text{Cl}_2 \)). A less reactive halogen cannot displace a more reactive one. So this reaction does not occur.
- **\( 2\text{LiI} + \text{Cl}_2
ightarrow 2\text{LiCl} + \text{I}_2 \)**
\( \text{Cl}_2 \) is more reactive than \( \text{I}_2 \) (since \( \text{Cl}_2 > \text{I}_2 \)). A more reactive halogen displaces a less reactive one. This reaction occurs, and products are more stable.
- **\( 2\text{LiCl} + \text{Br}_2
ightarrow 2\text{LiBr} + \text{Cl}_2 \)**
\( \text{Br}_2 \) is less reactive than \( \text{Cl}_2 \) (since \( \text{Cl}_2 > \text{Br}_2 \)). A less reactive halogen cannot displace a more reactive one. So this reaction does not occur.
- **\( 2\text{LiBr} + \text{F}_2
ightarrow 2\text{LiF} + \text{Br}_2 \)**
\( \text{F}_2 \) is more reactive than \( \text{Br}_2 \) (since \( \text{F}_2 > \text{Br}_2 \)). A more reactive halogen displaces a less reactive one. This reaction occurs, and products are more stable.
- **\( 2\text{LiBr} + \text{I}_2
ightarrow 2\text{LiI} + \text{Br}_2 \)**
\( \text{I}_2 \) is less reactive than \( \text{Br}_2 \) (since \( \text{Br}_2 > \text{I}_2 \)). A less reactive halogen cannot displace a more reactive one. So this reaction does not occur.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The correct reactions are \( 2\text{LiI} + \text{Cl}_2
ightarrow 2\text{LiCl} + \text{I}_2 \) and \( 2\text{LiBr} + \text{F}_2
ightarrow 2\text{LiF} + \text{Br}_2 \) (corresponding to the second and fourth options in the list).