QUESTION IMAGE
Question
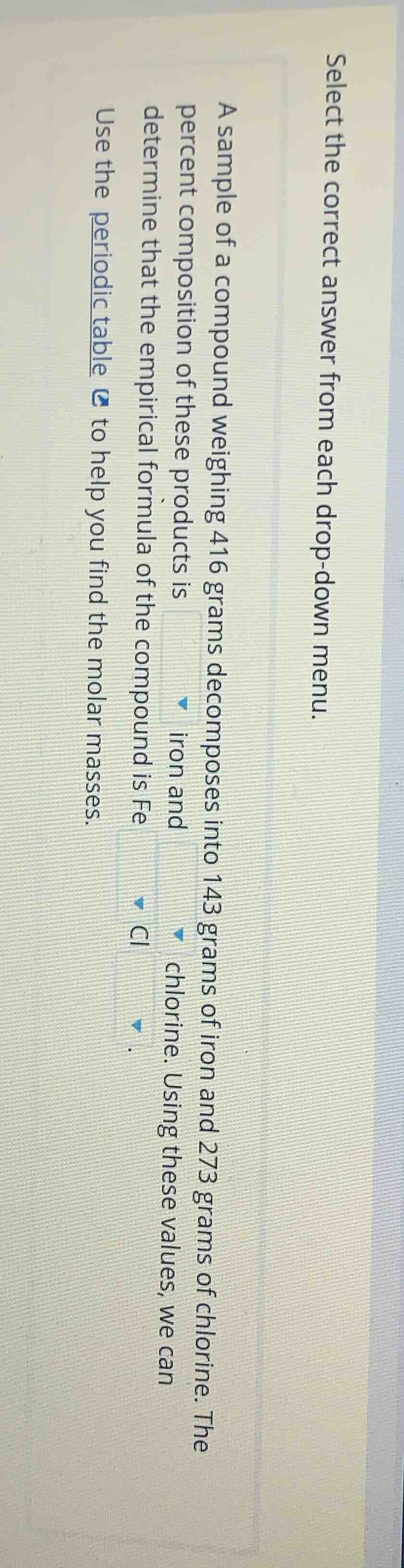
select the correct answer from each drop - down menu.
a sample of a compound weighing 416 grams decomposes into 143 grams of iron and 273 grams of chlorine. the percent composition of these products is
iron and
chlorine. using these values, we can determine that the empirical formula of the compound is fe
cl
.
use the periodic table to help you find the molar masses.
Step1: Find moles of Fe
Molar mass of Fe is \(55.85\space g/mol\). Moles of Fe = \(\frac{143\space g}{55.85\space g/mol} \approx 2.56\space mol\)
Step2: Find moles of Cl
Molar mass of Cl is \(35.45\space g/mol\). Moles of Cl = \(\frac{273\space g}{35.45\space g/mol} \approx 7.70\space mol\)
Step3: Find mole ratio
Divide moles by the smallest (2.56):
Fe: \(\frac{2.56}{2.56} = 1\)
Cl: \(\frac{7.70}{2.56} \approx 3\)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The empirical formula is \(FeCl_3\) (so the subscripts are 1 for Fe and 3 for Cl).