QUESTION IMAGE
Question
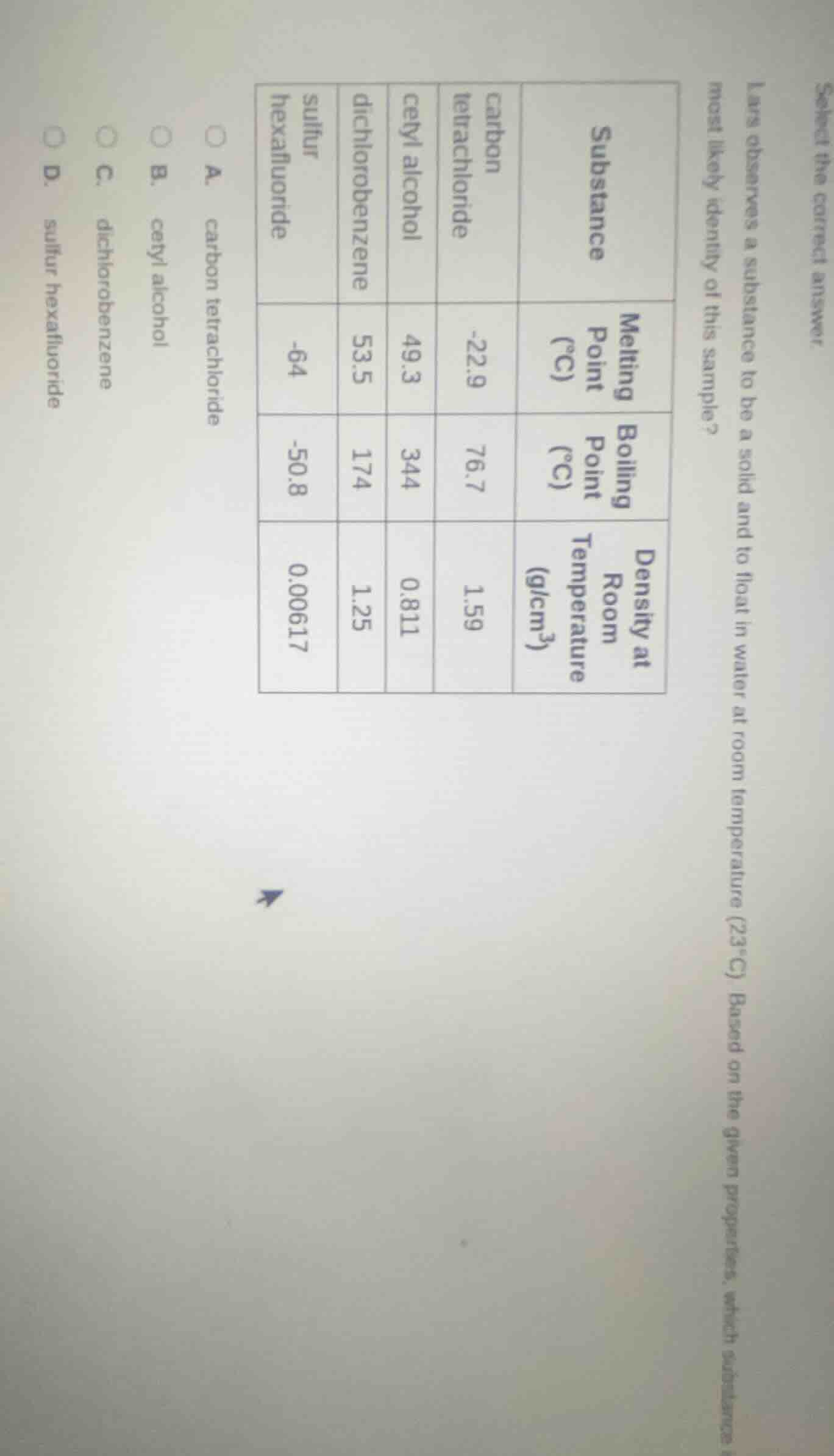
select the correct answer lars observes a substance to be a solid and to float in water at room temperature (23°c). based on the given properties, which substance is most likely identity of this sample?
substance | melting point (°c) | boiling point (°c) | density at room temperature (g/cm³)
carbon tetrachloride | -22.9 | 76.7 | 1.59
cetyl alcohol | 49.3 | 344 | 0.811
dichlorobenzene | 53.5 | 174 | 1.25
sulfur hexafluoride | -64 | -50.8 | 0.00617
a. carbon tetrachloride
b. cetyl alcohol
c. dichlorobenzene
d. sulfur hexafluoride
To determine the substance, we analyze two key properties: being a solid at room temperature (23°C) and having a density less than water (1 g/cm³, since it floats).
- State at 23°C: A substance is solid if its melting point (MP) > 23°C.
- Carbon tetrachloride: MP = -22.9°C (liquid at 23°C, eliminate A).
- Sulfur hexafluoride: MP = -64°C (gas/liquid at 23°C, eliminate D).
- Dichlorobenzene: MP = 53.5°C (solid at 23°C).
- Cetyl alcohol: MP = 49.3°C (solid at 23°C).
- Density (float in water): Density < 1 g/cm³.
- Dichlorobenzene: Density = 1.25 g/cm³ (>1, sinks, eliminate C).
- Cetyl alcohol: Density = 0.811 g/cm³ (<1, floats).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. cetyl alcohol