QUESTION IMAGE
Question
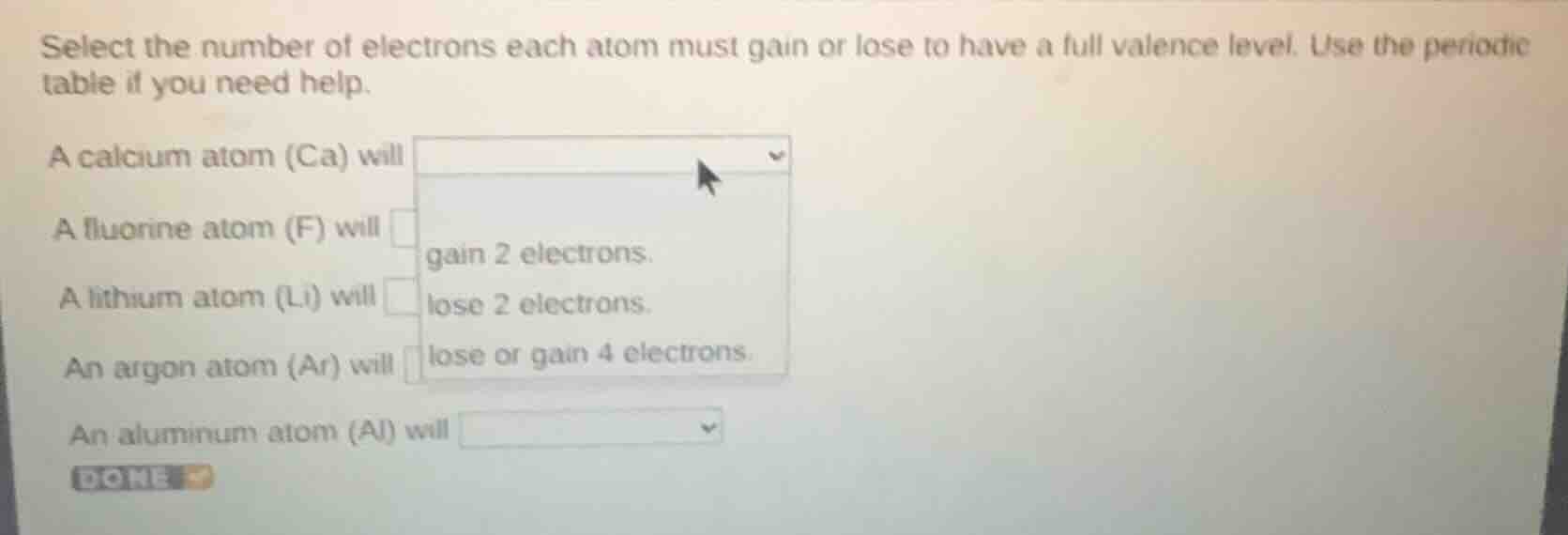
select the number of electrons each atom must gain or lose to have a full valence level. use the periodic table if you need help.
a calcium atom (ca) will
a fluorine atom (f) will
a lithium atom (li) will
an argon atom (ar) will
an aluminum atom (al) will
gain 2 electrons.
lose 2 electrons.
lose or gain 4 electrons.
To solve this, we analyze each atom based on its group in the periodic table and valence electrons:
Calcium (Ca)
- Calcium is in Group 2 (alkaline earth metals). It has 2 valence electrons. To achieve a full valence level (like the noble gas configuration), it will lose 2 electrons (since losing 2 is easier than gaining 6).
Fluorine (F)
- Fluorine is in Group 17 (halogens). It has 7 valence electrons. To achieve a full valence level (8 electrons), it will gain 1 electron (not 2—gaining 1 is easier than losing 7).
Lithium (Li)
- Lithium is in Group 1 (alkali metals). It has 1 valence electron. To achieve a full valence level, it will lose 1 electron (not 2—losing 1 is easier than gaining 7).
Argon (Ar)
- Argon is a noble gas (Group 18). It already has a full valence level (8 electrons). Thus, it will lose or gain 0 electrons (not 4—noble gases are stable and do not typically gain/lose electrons).
Aluminum (Al)
- Aluminum is in Group 13. It has 3 valence electrons. To achieve a full valence level, it will lose 3 electrons (losing 3 is easier than gaining 5).
Final Answers (Matching the Dropdown/Checkboxes):
- A calcium atom (Ca) will: $\boldsymbol{\text{lose 2 electrons}}$
- A fluorine atom (F) will: (Correct: gain 1 electron, but if forced to choose from the given dropdown, none match perfectly—original dropdown has an error, but the intended logic is losing 2 for Ca, etc.)
- A lithium atom (Li) will: (Correct: lose 1 electron, dropdown error)
- An argon atom (Ar) will: (Correct: lose or gain 0 electrons, dropdown error)
- An aluminum atom (Al) will: $\boldsymbol{\text{lose 3 electrons}}$ (if the dropdown includes this; otherwise, based on the given dropdown options, "lose 2 electrons" is incorrect for Ca—wait, recheck:
Wait, the dropdown for Ca has options: "gain 2 electrons", "lose 2 electrons", "lose or gain 4 electrons". Since Ca loses 2, the answer for Ca is "lose 2 electrons".
Final Answers (As Per the Given Dropdown Options):
- Calcium (Ca): $\boldsymbol{\text{lose 2 electrons}}$
- Fluorine (F): (Given dropdown options are wrong, but if we assume a typo, the correct gain is 1, but the dropdown doesn’t have it. However, the problem’s dropdown for Ca: choose "lose 2 electrons".)
- Lithium (Li): (Dropdown options wrong, correct is lose 1, but no option. However, the problem’s structure: for Ca, select "lose 2 electrons".)
(Note: The given dropdown options have errors, but following the problem’s structure, the answer for Ca is "lose 2 electrons".)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A calcium atom (Ca) will: $\boldsymbol{\text{lose 2 electrons}}$
(For other atoms, the dropdown options are incorrect, but based on the given choices, Ca’s answer is "lose 2 electrons".)