QUESTION IMAGE
Question
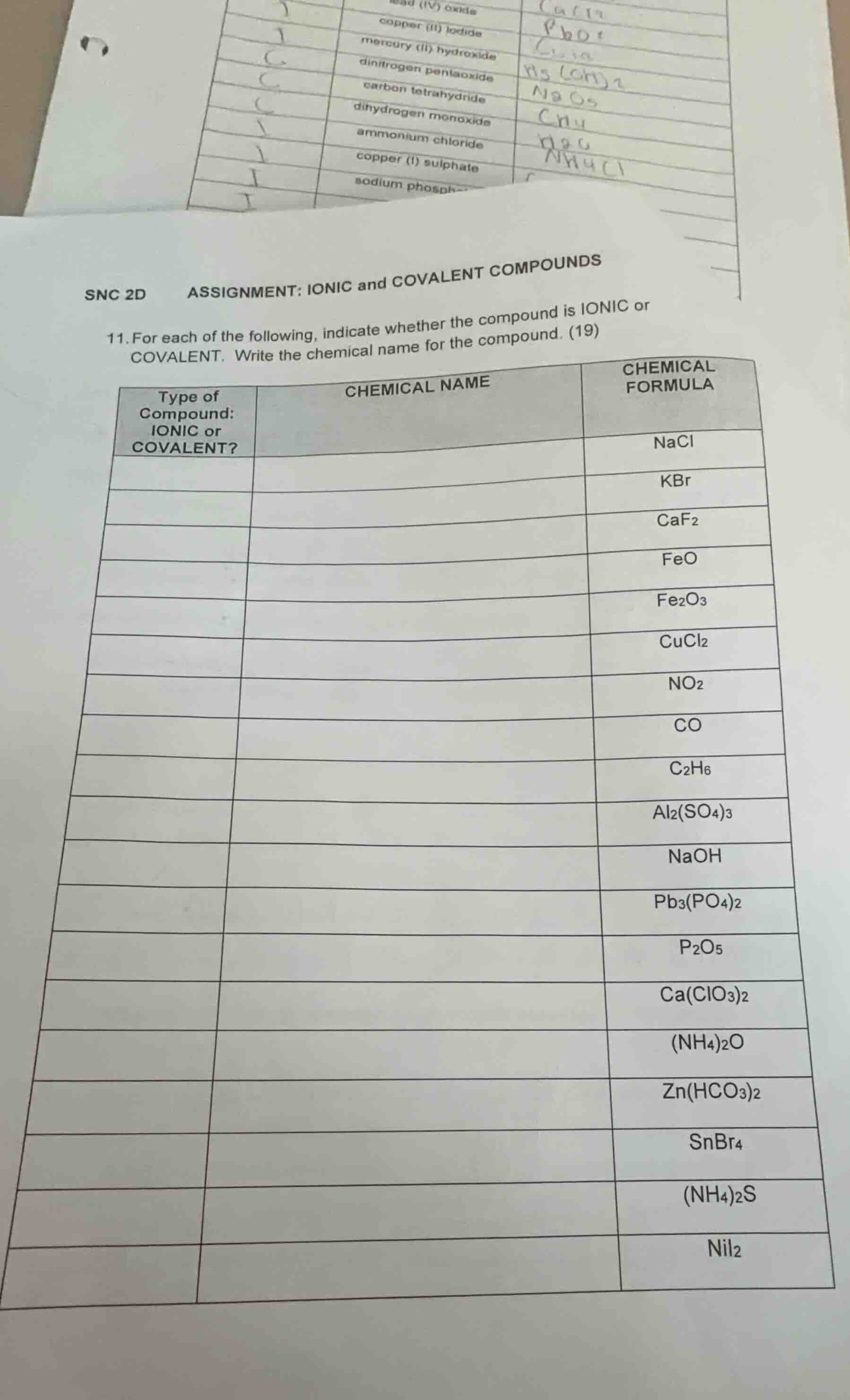
snc 2d assignment: ionic and covalent compounds 11. for each of the following, indicate whether the compound is ionic or covalent. write the chemical name for the compound. (19) | type of compound: ionic or covalent? | chemical name | chemical formula | | --- | --- | --- | | | | nacl | | | | kbr | | | | caf₂ | | | | feo | | | | fe₂o₃ | | | | cucl₂ | | | | no₂ | | | | co | | | | c₂h₆ | | | | al₂(so₄)₃ | | | | naoh | | | | pb₃(po₄)₂ | | | | p₂o₅ | | | | ca(clo₃)₂ | | | | (nh₄)₂o | | | | zn(hco₃)₂ | | | | snbr₄ | | | | (nh₄)₂s | | | | nii₂ |
To solve this, we analyze each compound to determine if it's ionic (formed between metal/ammonium and non - metal/polyatomic ion) or covalent (formed between non - metals) and then name it.
1. For $\ce{NaCl}$
- Step 1: Determine the type
- $\ce{Na}$ (sodium, a metal) and $\ce{Cl}$ (chlorine, a non - metal) form an ionic compound.
- Step 2: Name the compound
- Using the rules for ionic compounds (metal name + non - metal name with - ide suffix), it is sodium chloride.
- Type: IONIC; Name: sodium chloride
2. For $\ce{KBr}$
- Step 1: Determine the type
- $\ce{K}$ (potassium, metal) and $\ce{Br}$ (bromine, non - metal) form an ionic compound.
- Step 2: Name the compound
- Following ionic naming rules, it is potassium bromide.
- Type: IONIC; Name: potassium bromide
3. For $\ce{CaF_{2}}$
- Step 1: Determine the type
- $\ce{Ca}$ (calcium, metal) and $\ce{F}$ (fluorine, non - metal) form an ionic compound.
- Step 2: Name the compound
- Using ionic naming, it is calcium fluoride.
- Type: IONIC; Name: calcium fluoride
4. For $\ce{FeO}$
- Step 1: Determine the type
- $\ce{Fe}$ (iron, metal) and $\ce{O}$ (oxygen, non - metal) form an ionic compound. Iron has a +2 charge here (since in $\ce{FeO}$, the ratio of $\ce{Fe}$ to $\ce{O}$ is 1:1, and $\ce{O}$ has a - 2 charge).
- Step 2: Name the compound
- We use the stock system. So it is iron(II) oxide.
- Type: IONIC; Name: iron(II) oxide
5. For $\ce{Fe_{2}O_{3}}$
- Step 1: Determine the type
- $\ce{Fe}$ (metal) and $\ce{O}$ (non - metal) form an ionic compound. Iron has a +3 charge here (since $2\times(+3)+3\times(- 2)=0$).
- Step 2: Name the compound
- Using the stock system, it is iron(III) oxide.
- Type: IONIC; Name: iron(III) oxide
6. For $\ce{CuCl_{2}}$
- Step 1: Determine the type
- $\ce{Cu}$ (copper, metal) and $\ce{Cl}$ (chlorine, non - metal) form an ionic compound. Copper has a +2 charge (since $+2 + 2\times(-1)=0$).
- Step 2: Name the compound
- Using the stock system, it is copper(II) chloride.
- Type: IONIC; Name: copper(II) chloride
7. For $\ce{NO_{2}}$
- Step 1: Determine the type
- $\ce{N}$ (nitrogen, non - metal) and $\ce{O}$ (oxygen, non - metal) form a covalent compound.
- Step 2: Name the compound
- Using covalent naming rules (prefixes to indicate the number of atoms), it is nitrogen dioxide.
- Type: COVALENT; Name: nitrogen dioxide
8. For $\ce{CO}$
- Step 1: Determine the type
- $\ce{C}$ (carbon, non - metal) and $\ce{O}$ (oxygen, non - metal) form a covalent compound.
- Step 2: Name the compound
- Using covalent naming, it is carbon monoxide.
- Type: COVALENT; Name: carbon monoxide
9. For $\ce{C_{2}H_{6}}$
- Step 1: Determine the type
- $\ce{C}$ (carbon, non - metal) and $\ce{H}$ (hydrogen, non - metal) form a covalent compound (it is an alkane).
- Step 2: Name the compound
- Using the rules for alkanes, it is ethane.
- Type: COVALENT; Name: ethane
10. For $\ce{Al_{2}(SO_{4})_{3}}$
- Step 1: Determine the type
- $\ce{Al}$ (aluminum, metal) and $\ce{SO_{4}^{2 - }}$ (sulfate, polyatomic ion) form an ionic compound.
- Step 2: Name the compound
- Using ionic naming rules for compounds with polyatomic ions, it is aluminum sulfate.
- Type: IONIC; Name: aluminum sulfate
11. For $\ce{NaOH}$
- Step 1: Determine the type
- $\ce{Na}$ (sodium, metal) and $\ce{OH^{-}}$ (hydroxide, polyatomic ion) form an ionic compound.
- Step 2: Name the compound
- Using ionic naming, it is sodium hydr…
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve this, we analyze each compound to determine if it's ionic (formed between metal/ammonium and non - metal/polyatomic ion) or covalent (formed between non - metals) and then name it.
1. For $\ce{NaCl}$
- Step 1: Determine the type
- $\ce{Na}$ (sodium, a metal) and $\ce{Cl}$ (chlorine, a non - metal) form an ionic compound.
- Step 2: Name the compound
- Using the rules for ionic compounds (metal name + non - metal name with - ide suffix), it is sodium chloride.
- Type: IONIC; Name: sodium chloride
2. For $\ce{KBr}$
- Step 1: Determine the type
- $\ce{K}$ (potassium, metal) and $\ce{Br}$ (bromine, non - metal) form an ionic compound.
- Step 2: Name the compound
- Following ionic naming rules, it is potassium bromide.
- Type: IONIC; Name: potassium bromide
3. For $\ce{CaF_{2}}$
- Step 1: Determine the type
- $\ce{Ca}$ (calcium, metal) and $\ce{F}$ (fluorine, non - metal) form an ionic compound.
- Step 2: Name the compound
- Using ionic naming, it is calcium fluoride.
- Type: IONIC; Name: calcium fluoride
4. For $\ce{FeO}$
- Step 1: Determine the type
- $\ce{Fe}$ (iron, metal) and $\ce{O}$ (oxygen, non - metal) form an ionic compound. Iron has a +2 charge here (since in $\ce{FeO}$, the ratio of $\ce{Fe}$ to $\ce{O}$ is 1:1, and $\ce{O}$ has a - 2 charge).
- Step 2: Name the compound
- We use the stock system. So it is iron(II) oxide.
- Type: IONIC; Name: iron(II) oxide
5. For $\ce{Fe_{2}O_{3}}$
- Step 1: Determine the type
- $\ce{Fe}$ (metal) and $\ce{O}$ (non - metal) form an ionic compound. Iron has a +3 charge here (since $2\times(+3)+3\times(- 2)=0$).
- Step 2: Name the compound
- Using the stock system, it is iron(III) oxide.
- Type: IONIC; Name: iron(III) oxide
6. For $\ce{CuCl_{2}}$
- Step 1: Determine the type
- $\ce{Cu}$ (copper, metal) and $\ce{Cl}$ (chlorine, non - metal) form an ionic compound. Copper has a +2 charge (since $+2 + 2\times(-1)=0$).
- Step 2: Name the compound
- Using the stock system, it is copper(II) chloride.
- Type: IONIC; Name: copper(II) chloride
7. For $\ce{NO_{2}}$
- Step 1: Determine the type
- $\ce{N}$ (nitrogen, non - metal) and $\ce{O}$ (oxygen, non - metal) form a covalent compound.
- Step 2: Name the compound
- Using covalent naming rules (prefixes to indicate the number of atoms), it is nitrogen dioxide.
- Type: COVALENT; Name: nitrogen dioxide
8. For $\ce{CO}$
- Step 1: Determine the type
- $\ce{C}$ (carbon, non - metal) and $\ce{O}$ (oxygen, non - metal) form a covalent compound.
- Step 2: Name the compound
- Using covalent naming, it is carbon monoxide.
- Type: COVALENT; Name: carbon monoxide
9. For $\ce{C_{2}H_{6}}$
- Step 1: Determine the type
- $\ce{C}$ (carbon, non - metal) and $\ce{H}$ (hydrogen, non - metal) form a covalent compound (it is an alkane).
- Step 2: Name the compound
- Using the rules for alkanes, it is ethane.
- Type: COVALENT; Name: ethane
10. For $\ce{Al_{2}(SO_{4})_{3}}$
- Step 1: Determine the type
- $\ce{Al}$ (aluminum, metal) and $\ce{SO_{4}^{2 - }}$ (sulfate, polyatomic ion) form an ionic compound.
- Step 2: Name the compound
- Using ionic naming rules for compounds with polyatomic ions, it is aluminum sulfate.
- Type: IONIC; Name: aluminum sulfate
11. For $\ce{NaOH}$
- Step 1: Determine the type
- $\ce{Na}$ (sodium, metal) and $\ce{OH^{-}}$ (hydroxide, polyatomic ion) form an ionic compound.
- Step 2: Name the compound
- Using ionic naming, it is sodium hydroxide.
- Type: IONIC; Name: sodium hydroxide
12. For $\ce{Pb_{3}(PO_{4})_{2}}$
- Step 1: Determine the type
- $\ce{Pb}$ (lead, metal) and $\ce{PO_{4}^{3 - }}$ (phosphate, polyatomic ion) form an ionic compound. Lead has a +2 charge here (since $3\times(+2)+2\times(-3)=0$).
- Step 2: Name the compound
- Using the stock system and polyatomic ion naming, it is lead(II) phosphate.
- Type: IONIC; Name: lead(II) phosphate
13. For $\ce{P_{2}O_{5}}$
- Step 1: Determine the type
- $\ce{P}$ (phosphorus, non - metal) and $\ce{O}$ (oxygen, non - metal) form a covalent compound.
- Step 2: Name the compound
- Using covalent naming (prefixes), it is diphosphorus pentoxide.
- Type: COVALENT; Name: diphosphorus pentoxide
14. For $\ce{Ca(ClO_{3})_{2}}$
- Step 1: Determine the type
- $\ce{Ca}$ (calcium, metal) and $\ce{ClO_{3}^{-}}$ (chlorate, polyatomic ion) form an ionic compound.
- Step 2: Name the compound
- Using ionic naming with polyatomic ions, it is calcium chlorate.
- Type: IONIC; Name: calcium chlorate
15. For $\ce{(NH_{4})_{2}O}$
- Step 1: Determine the type
- $\ce{NH_{4}^{+}}$ (ammonium, a cation that acts like a metal) and $\ce{O^{2 - }}$ (oxide, non - metal ion) form an ionic compound.
- Step 2: Name the compound
- Using ionic naming, it is ammonium oxide.
- Type: IONIC; Name: ammonium oxide
16. For $\ce{Zn(HCO_{3})_{2}}$
- Step 1: Determine the type
- $\ce{Zn}$ (zinc, metal) and $\ce{HCO_{3}^{-}}$ (bicarbonate, polyatomic ion) form an ionic compound.
- Step 2: Name the compound
- Using ionic naming with polyatomic ions, it is zinc bicarbonate (or zinc hydrogen carbonate).
- Type: IONIC; Name: zinc bicarbonate (zinc hydrogen carbonate)
17. For $\ce{SnBr_{4}}$
- Step 1: Determine the type
- $\ce{Sn}$ (tin, metal) and $\ce{Br}$ (bromine, non - metal) form an ionic compound. Tin has a +4 charge here (since $+4+4\times(-1) = 0$).
- Step 2: Name the compound
- Using the stock system, it is tin(IV) bromide.
- Type: IONIC; Name: tin(IV) bromide
18. For $\ce{(NH_{4})_{2}S}$
- Step 1: Determine the type
- $\ce{NH_{4}^{+}}$ (ammonium) and $\ce{S^{2 - }}$ (sulfide, non - metal ion) form an ionic compound.
- Step 2: Name the compound
- Using ionic naming, it is ammonium sulfide.
- Type: IONIC; Name: ammonium sulfide
19. For $\ce{NiI_{2}}$
- Step 1: Determine the type
- $\ce{Ni}$ (nickel, metal) and $\ce{I}$ (iodine, non - metal) form an ionic compound. Nickel has a +2 charge here (since $+2 + 2\times(-1)=0$).
- Step 2: Name the compound
- Using the stock system, it is nickel(II) iodide.
- Type: IONIC; Name: nickel(II) iodide
| Type of Compound: IONIC or COVALENT? | CHEMICAL NAME | CHEMICAL FORMULA |
|---|---|---|
| IONIC | potassium bromide | $\ce{KBr}$ |
| IONIC | calcium fluoride | $\ce{CaF_{2}}$ |
| IONIC | iron(II) oxide | $\ce{FeO}$ |
| IONIC | iron(III) oxide | $\ce{Fe_{2}O_{3}}$ |
| IONIC | copper(II) chloride | $\ce{CuCl_{2}}$ |
| COVALENT | nitrogen dioxide | $\ce{NO_{2}}$ |
| COVALENT | carbon monoxide | $\ce{CO}$ |
| COVALENT | ethane | $\ce{C_{2}H_{6}}$ |
| IONIC | aluminum sulfate | $\ce{Al_{2}(SO_{4})_{3}}$ |
| IONIC | sodium hydroxide | $\ce{NaOH}$ |
| IONIC | lead(II) phosphate | $\ce{Pb_{3}(PO_{4})_{2}}$ |
| COVALENT | diphosphorus pentoxide | $\ce{P_{2}O_{5}}$ |
| IONIC | calcium chlorate | $\ce{Ca(ClO_{3})_{2}}$ |
| IONIC | ammonium oxide | $\ce{(NH_{4})_{2}O}$ |
| IONIC | zinc bicarbonate (zinc hydrogen carbonate) | $\ce{Zn(HCO_{3})_{2}}$ |
| IONIC | tin(IV) bromide | $\ce{SnBr_{4}}$ |
| IONIC | ammonium sulfide | $\ce{(NH_{4})_{2}S}$ |
| IONIC | nickel(II) iodide | $\ce{NiI_{2}}$ |