QUESTION IMAGE
Question
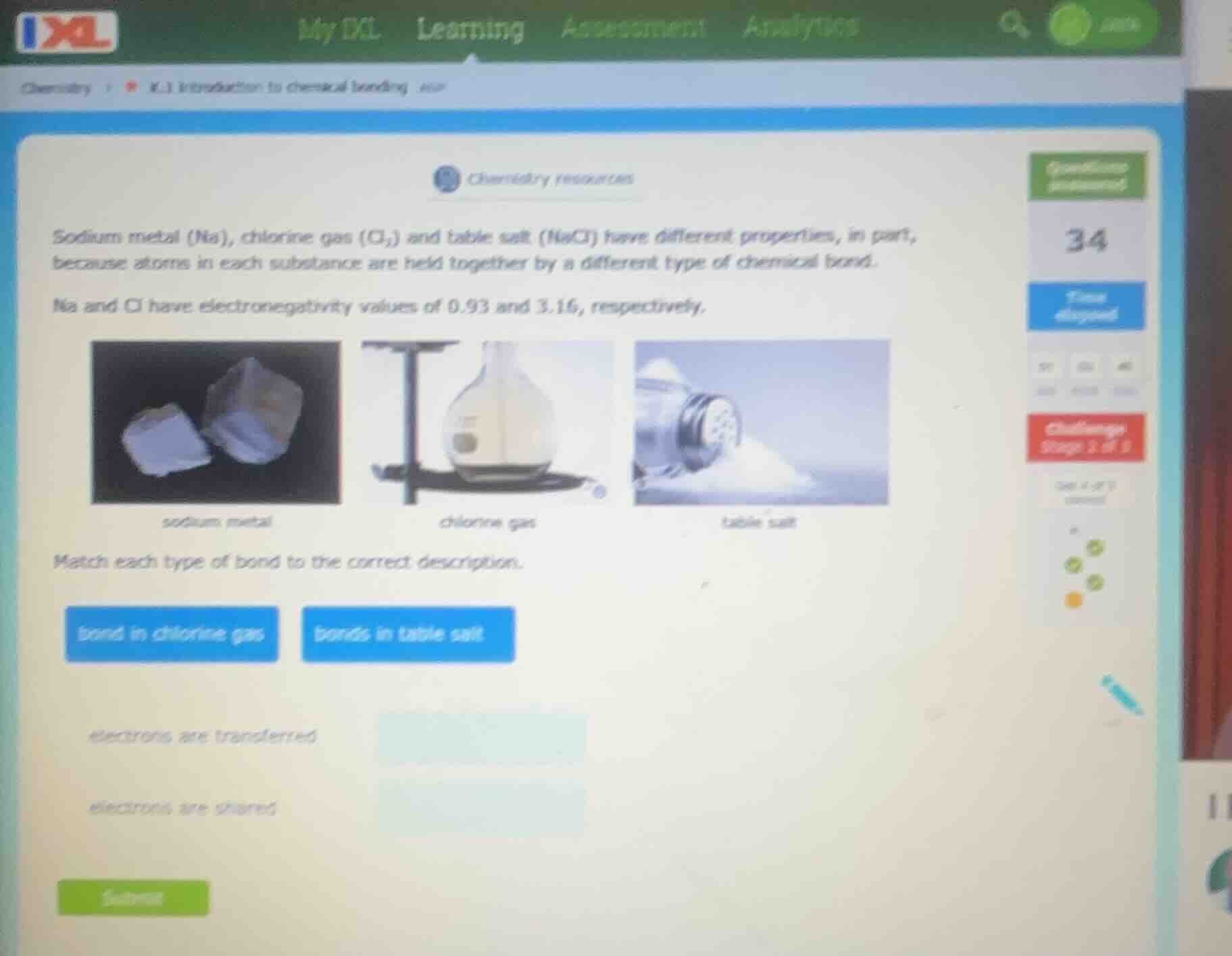
sodium metal (na), chlorine gas ($cl_2$) and table salt (nacl) have different properties, in part, because atoms in each substance are held together by a different type of chemical bond.
na and cl have electronegativity values of 0.93 and 3.16, respectively.
match each type of bond to the correct description.
bond in chlorine gas
bonds in table salt
electrons are transferred
electrons are shared
Brief Explanations
- Table salt (NaCl) forms between a metal (Na) and nonmetal (Cl) with a large electronegativity difference ($3.16 - 0.93 = 2.23$). This large difference causes electron transfer, forming an ionic bond.
- Chlorine gas ($Cl_2$) consists of two identical nonmetal atoms. Their equal electronegativity means electrons are shared equally in a covalent bond.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- electrons are transferred: bonds in table salt
- electrons are shared: bond in chlorine gas