QUESTION IMAGE
Question
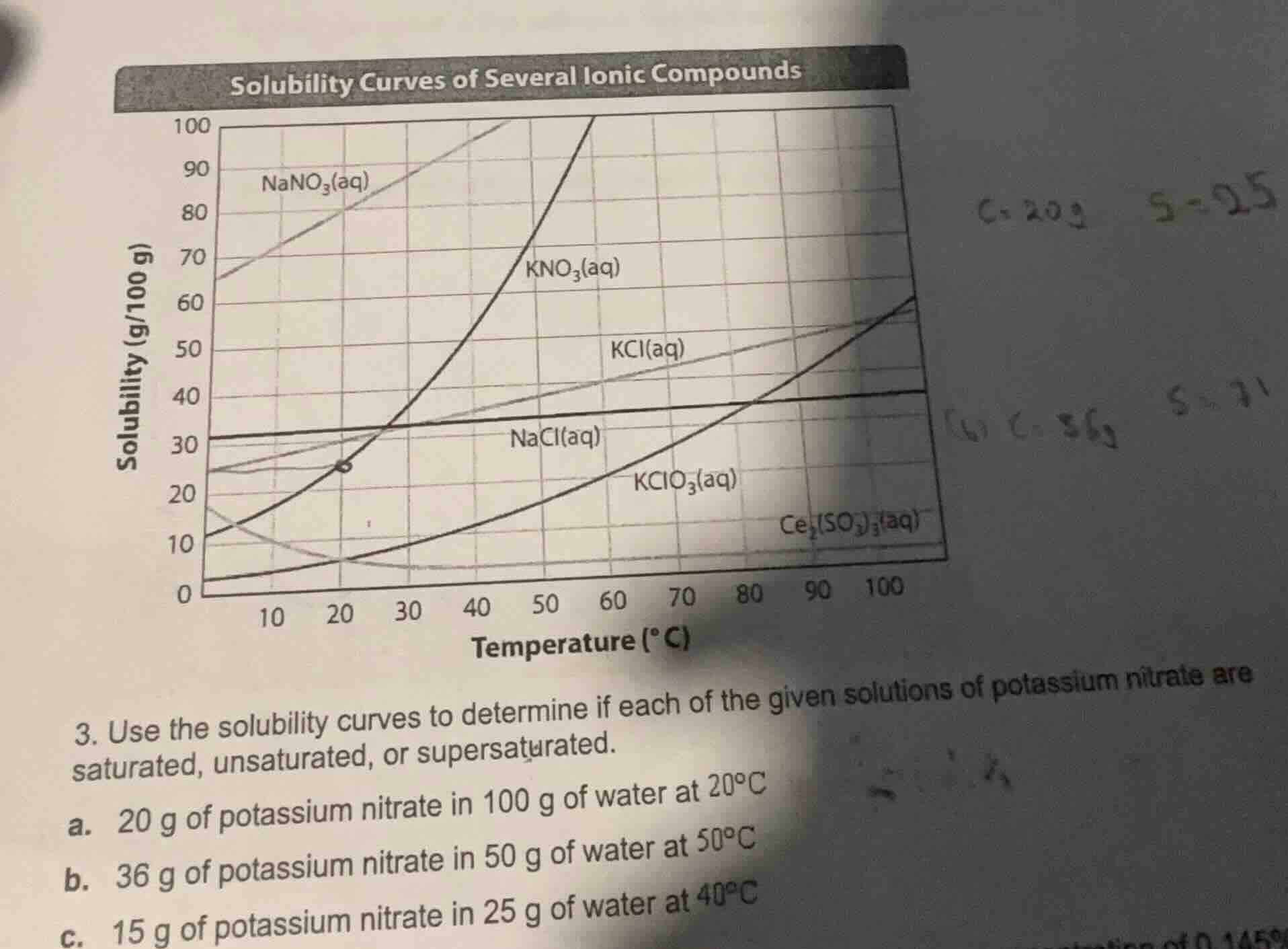
solubility curves of several ionic compounds
- use the solubility curves to determine if each of the given solutions of potassium nitrate are saturated, unsaturated, or supersaturated.
a. 20 g of potassium nitrate in 100 g of water at 20°c
b. 36 g of potassium nitrate in 50 g of water at 50°c
c. 15 g of potassium nitrate in 25 g of water at 40°c
Part a: 20 g of \( \text{KNO}_3 \) in 100 g water at \( 20^\circ\text{C} \)
Step1: Find solubility of \( \text{KNO}_3 \) at \( 20^\circ\text{C} \)
From the solubility curve, at \( 20^\circ\text{C} \), the solubility of \( \text{KNO}_3 \) is about 25 g/100 g water.
Step2: Compare given amount with solubility
Given amount: 20 g/100 g water. Since \( 20 \, \text{g} < 25 \, \text{g} \), the solution is unsaturated.
Part b: 36 g of \( \text{KNO}_3 \) in 50 g water at \( 50^\circ\text{C} \)
Step1: Find solubility of \( \text{KNO}_3 \) at \( 50^\circ\text{C} \)
From the curve, at \( 50^\circ\text{C} \), solubility of \( \text{KNO}_3 \) is about 90 g/100 g water. For 50 g water, the solubility is \( \frac{90}{2} = 45 \, \text{g} \) (since solubility is per 100 g water, half the water means half the solubility).
Step2: Compare given amount with solubility
Given amount: 36 g in 50 g water. Since \( 36 \, \text{g} < 45 \, \text{g} \), the solution is unsaturated.
Part c: 15 g of \( \text{KNO}_3 \) in 25 g water at \( 40^\circ\text{C} \)
Step1: Find solubility of \( \text{KNO}_3 \) at \( 40^\circ\text{C} \)
From the curve, at \( 40^\circ\text{C} \), solubility of \( \text{KNO}_3 \) is about 64 g/100 g water. For 25 g water, solubility is \( \frac{64}{4} = 16 \, \text{g} \) (25 g is \( \frac{1}{4} \) of 100 g, so solubility is \( \frac{1}{4} \) of 64 g).
Step2: Compare given amount with solubility
Given amount: 15 g in 25 g water. Since \( 15 \, \text{g} < 16 \, \text{g} \), the solution is unsaturated.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
s:
a. Unsaturated
b. Unsaturated
c. Unsaturated