QUESTION IMAGE
Question
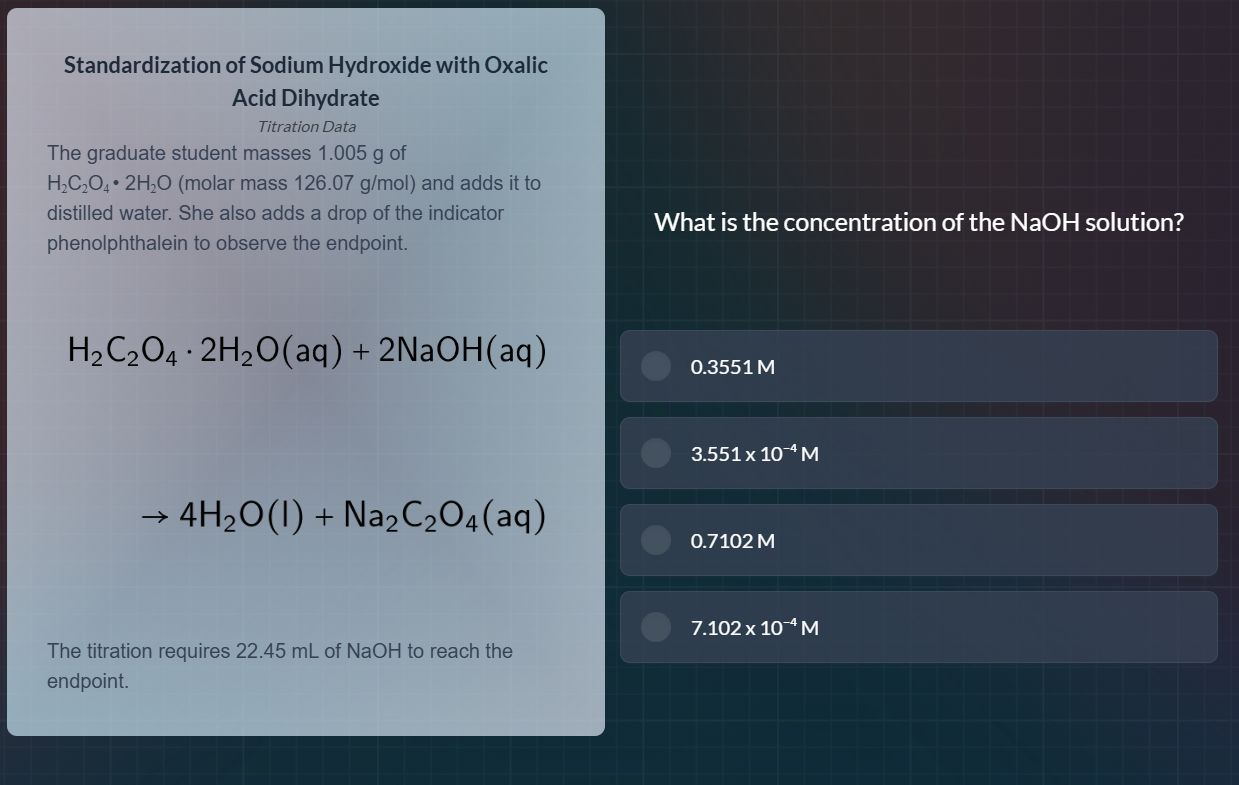
standardization of sodium hydroxide with oxalic acid dihydrate
titration data
the graduate student masses 1.005 g of
h₂c₂o₄·2h₂o (molar mass 126.07 g/mol) and adds it to
distilled water. she also adds a drop of the indicator
phenolphthalein to observe the endpoint.
h₂c₂o₄·2h₂o(aq) + 2naoh(aq)
→ 4h₂o(l) + na₂c₂o₄(aq)
the titration requires 22.45 ml of naoh to reach the
endpoint.
what is the concentration of the naoh solution?
0.3551 m
3.551 x 10⁻⁴ m
0.7102 m
7.102 x 10⁻⁴ m
Step1: Calculate moles of oxalic acid dihydrate
Moles = mass / molar mass. Mass of $\ce{H2C2O4\cdot 2H2O}$ is 1.005 g, molar mass is 126.07 g/mol.
$n(\ce{H2C2O4\cdot 2H2O}) = \frac{1.005\ \text{g}}{126.07\ \text{g/mol}} \approx 0.007972\ \text{mol}$
Step2: Determine moles of NaOH from stoichiometry
From the reaction: 1 mol $\ce{H2C2O4\cdot 2H2O}$ reacts with 2 mol NaOH.
$n(\ce{NaOH}) = 2 \times n(\ce{H2C2O4\cdot 2H2O}) = 2 \times 0.007972\ \text{mol} = 0.015944\ \text{mol}$
Step3: Calculate concentration of NaOH
Volume of NaOH is 22.45 mL = 0.02245 L. Molarity $M = \frac{n}{V}$.
$M(\ce{NaOH}) = \frac{0.015944\ \text{mol}}{0.02245\ \text{L}} \approx 0.7102\ \text{M}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
0.7102 M