QUESTION IMAGE
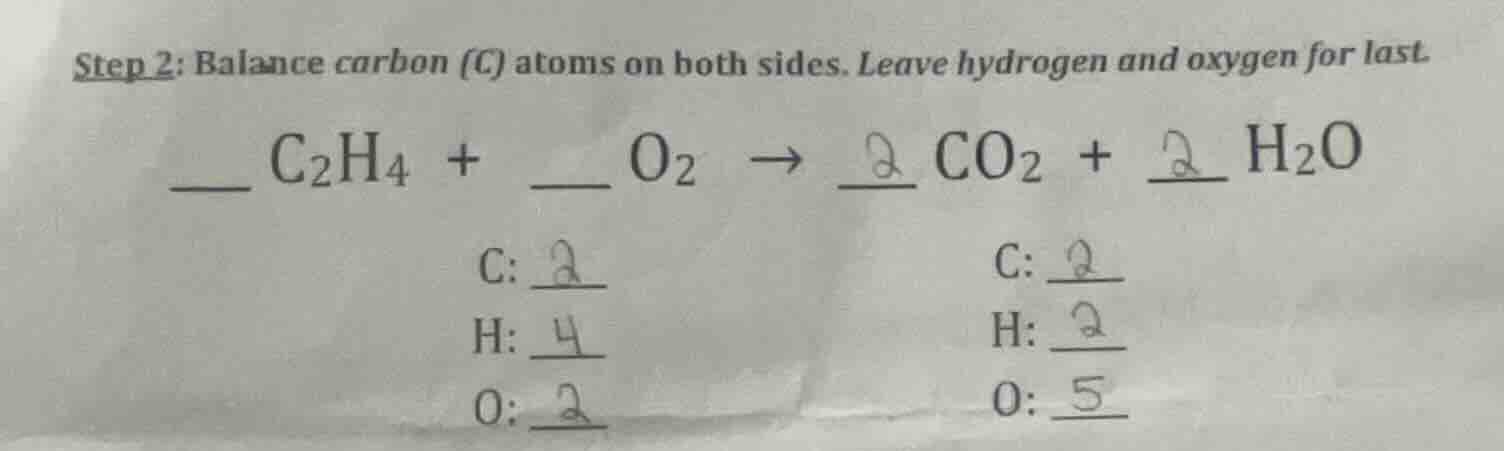
Question
step 2: balance carbon (c) atoms on both sides. leave hydrogen and oxygen for last. \underline{\quad} c₂h₄ + \underline{\quad} o₂ → \underline{2} co₂ + \underline{2} h₂o \qquad \qquad \qquad c: \underline{2} \qquad \qquad \qquad c: \underline{2} \qquad \qquad \qquad h: \underline{4} \qquad \qquad \qquad h: \underline{2} \qquad \qquad \qquad o: \underline{2} \qquad \qquad \qquad o: \underline{5}
Step1: Analyze Carbon Atoms
In \( \ce{C2H4} \), there are 2 C atoms. So, to balance C, we need 2 \( \ce{CO2} \) (since each \( \ce{CO2} \) has 1 C). So coefficient of \( \ce{CO2} \) is 2. Now C on left: 2 (from \( \ce{C2H4} \)), C on right: 2 (from \( 2\ce{CO2} \)) – balanced.
Step2: Analyze Hydrogen Atoms
In \( \ce{C2H4} \), there are 4 H atoms. Each \( \ce{H2O} \) has 2 H, so we need \( \frac{4}{2}=2 \) \( \ce{H2O} \). So coefficient of \( \ce{H2O} \) is 2. Now H on left: 4 (from \( \ce{C2H4} \)), H on right: \( 2\times2 = 4 \) (from \( 2\ce{H2O} \)) – balanced.
Step3: Analyze Oxygen Atoms
On right: \( 2\ce{CO2} \) has \( 2\times2 = 4 \) O, \( 2\ce{H2O} \) has \( 2\times1 = 2 \) O. Total O on right: \( 4 + 2 = 6 \). Each \( \ce{O2} \) has 2 O, so coefficient of \( \ce{O2} \) is \( \frac{6}{2}=3 \). Now O on left: \( 3\times2 = 6 \), O on right: 6 – balanced.
Now, the balanced equation is \( \ce{C2H4 + 3O2 -> 2CO2 + 2H2O} \).
For the atom counts:
- Left ( \( \ce{C2H4 + 3O2} \) ):
- C: 2 (from \( \ce{C2H4} \))
- H: 4 (from \( \ce{C2H4} \))
- O: \( 3\times2 = 6 \) (from \( 3\ce{O2} \))
- Right ( \( 2\ce{CO2} + 2\ce{H2O} \) ):
- C: \( 2\times1 = 2 \) (from \( 2\ce{CO2} \))
- H: \( 2\times2 = 4 \) (from \( 2\ce{H2O} \))
- O: \( (2\times2)+(2\times1)=4 + 2 = 6 \) (from \( 2\ce{CO2} \) and \( 2\ce{H2O} \))
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Balanced equation: \( \boldsymbol{\ce{C2H4 + 3O2 -> 2CO2 + 2H2O}} \)
Atom counts (left/right):
C: 2/2, H: 4/4, O: 6/6