QUESTION IMAGE
Question
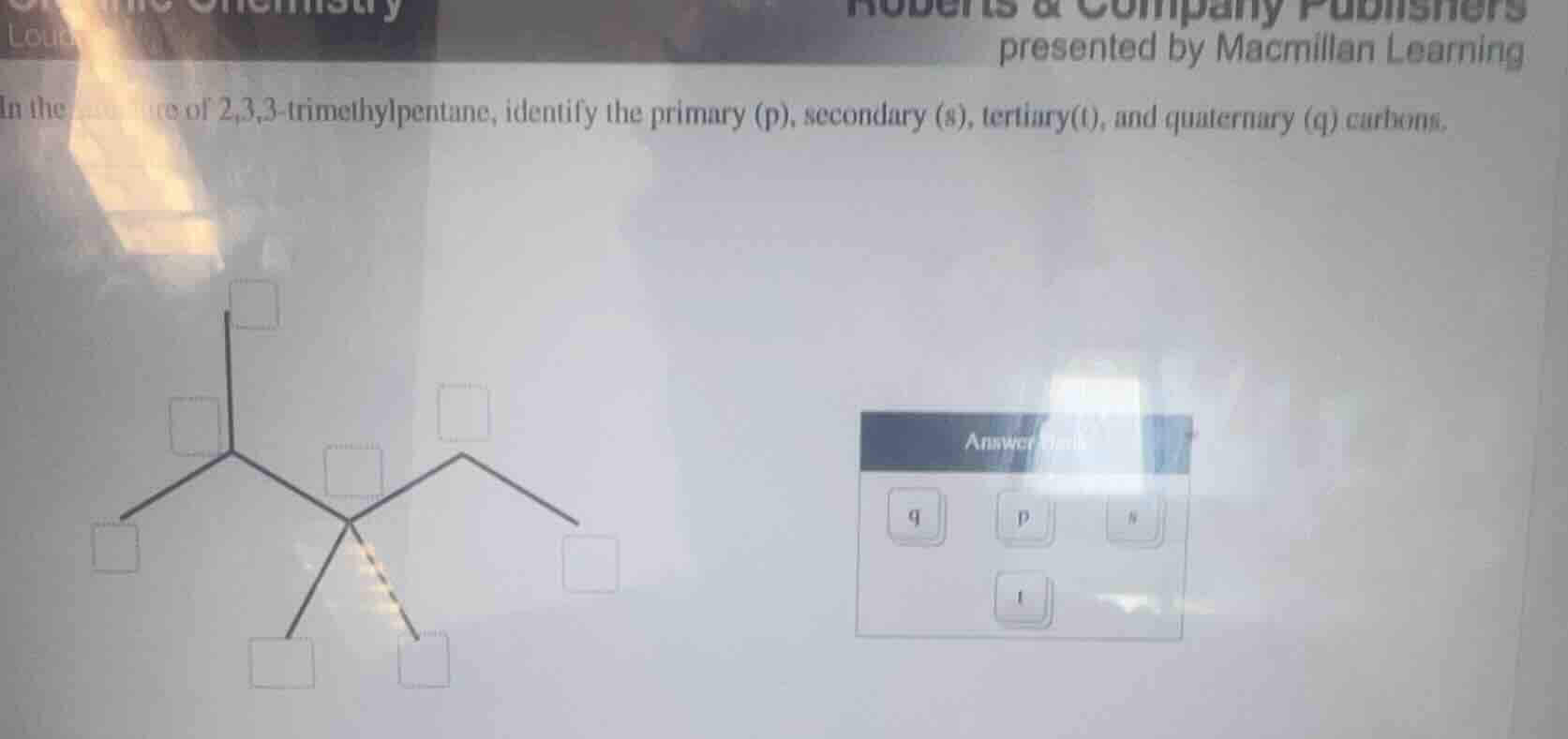
in the structure of 2,3,3 - trimethylpentane, identify the primary (p), secondary (s), tertiary(t), and quaternary (q) carbons.
Brief Explanations
- Primary (p) carbons: Bonded to only 1 other carbon; these are all the end (terminal) methyl carbons. There are 5 of these.
- Secondary (s) carbon: Bonded to 2 other carbons; this is the CH₂ carbon on the right side of the main chain. There is 1 of these.
- Tertiary (t) carbon: Bonded to 3 other carbons; this is the CH carbon on the left side of the main chain (carbon 2). There is 1 of these.
- Quaternary (q) carbon: Bonded to 4 other carbons; this is the central carbon with four carbon branches (carbon 3). There is 1 of these.
To map to the structure:
- The 5 terminal (outermost) empty boxes = p
- The CH₂ box (right side, middle chain) = s
- The CH box (left side, connected to 3 carbons) = t
- The central box connected to 4 carbons = q
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- 5 terminal carbons: p
- Central 4-bonded carbon: q
- CH carbon (left middle): t
- CH₂ carbon (right middle): s