QUESTION IMAGE
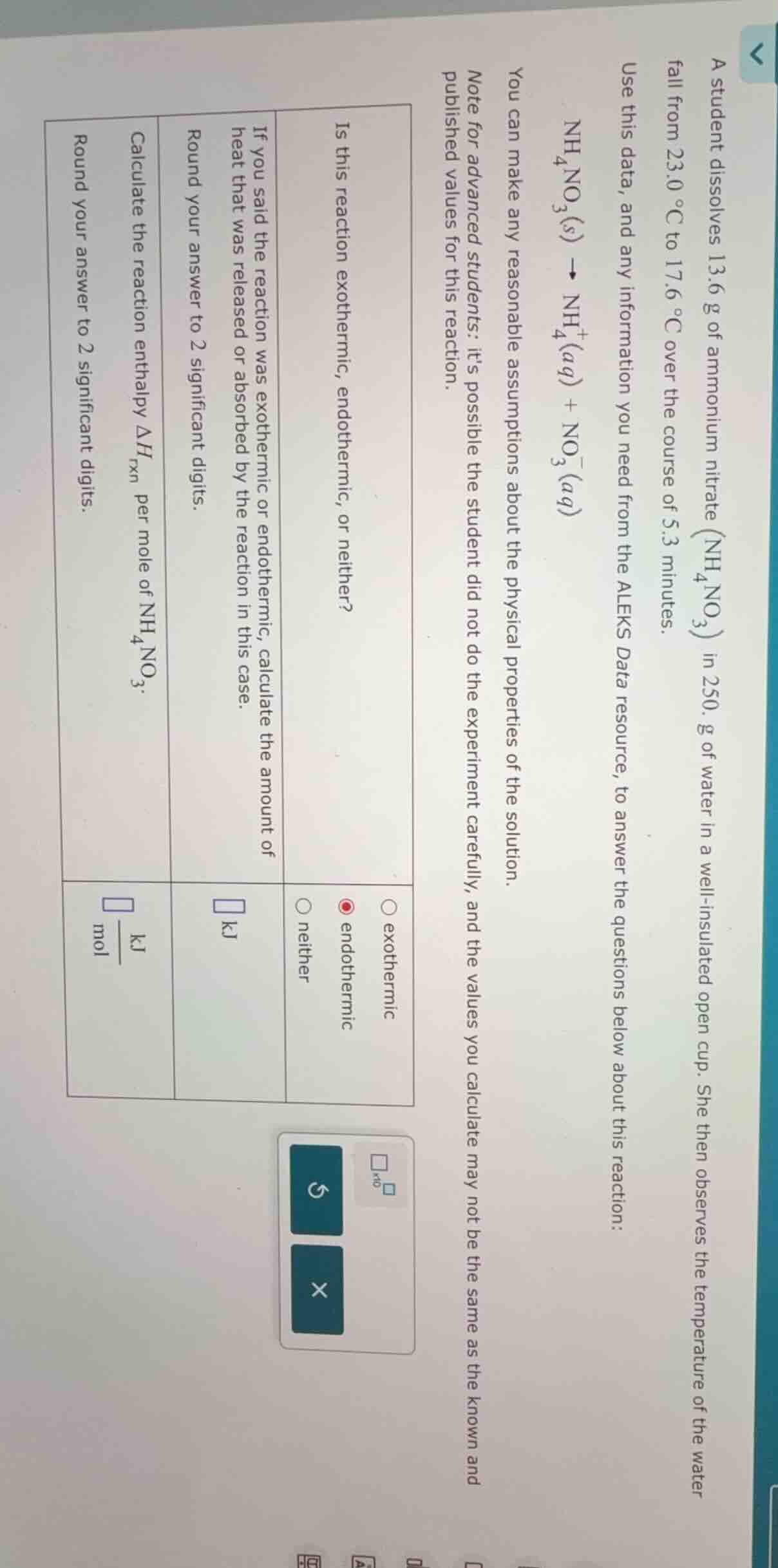
Question
a student dissolves 13.6 g of ammonium nitrate ($\text{nh}_4\text{no}_3$) in 250. g of water in a well-insulated open cup. she then observes the temperature of the water fall from 23.0 °c to 17.6 °c over the course of 5.3 minutes.
use this data, and any information you need from the aleks data resource, to answer the questions below about this reaction:
$\text{nh}_4\text{no}_3(s) \
ightarrow \text{nh}_4^+(aq) + \text{no}_3^-(aq)$
you can make any reasonable assumptions about the physical properties of the solution.
note for advanced students: its possible the student did not do the experiment carefully, and the values you calculate may not be the same as the known and published values for this reaction.
- is this reaction exothermic, endothermic, or neither?
○ exothermic
● endothermic
○ neither
- if you said the reaction was exothermic or endothermic, calculate the amount of heat that was released or absorbed by the reaction in this case.
round your answer to 2 significant digits.
$\square$ kj
- calculate the reaction enthalpy $\delta h_{\text{rxn}}$ per mole of $\text{nh}_4\text{no}_3$.
round your answer to 2 significant digits.
$\square$ $\frac{\text{kj}}{\text{mol}}$
Step1: Identify reaction type
The water temperature decreases, so the reaction absorbs heat from the surroundings, meaning it is endothermic.
Step2: Calculate heat change of solution
Assume the solution has the same specific heat as water ($c = 4.184\ \frac{\text{J}}{\text{g}^\circ\text{C}}$) and total mass is $250. + 13.6 = 263.6\ \text{g}$.
Temperature change: $\Delta T = 17.6^\circ\text{C} - 23.0^\circ\text{C} = -5.4^\circ\text{C}$
Heat change of solution: $q_{\text{soln}} = mc\Delta T = 263.6\ \text{g} \times 4.184\ \frac{\text{J}}{\text{g}^\circ\text{C}} \times (-5.4^\circ\text{C}) = -5980\ \text{J} = -5.98\ \text{kJ}$
Step3: Find heat absorbed by reaction
For an insulated system, $q_{\text{rxn}} = -q_{\text{soln}} = -(-5.98\ \text{kJ}) = 6.0\ \text{kJ}$ (rounded to 2 sig figs)
Step4: Calculate moles of $\text{NH}_4\text{NO}_3$
Molar mass of $\text{NH}_4\text{NO}_3$: $14.01 + 4\times1.008 + 14.01 + 3\times16.00 = 80.052\ \frac{\text{g}}{\text{mol}}$
Moles: $n = \frac{13.6\ \text{g}}{80.052\ \frac{\text{g}}{\text{mol}}} = 0.1699\ \text{mol}$
Step5: Calculate $\Delta H_{\text{rxn}}$ per mole
$\Delta H_{\text{rxn}} = \frac{q_{\text{rxn}}}{n} = \frac{6.0\ \text{kJ}}{0.1699\ \text{mol}} = 35\ \frac{\text{kJ}}{\text{mol}}$ (rounded to 2 sig figs)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Is this reaction exothermic, endothermic, or neither?
endothermic
Amount of heat absorbed by the reaction:
6.0 kJ
Reaction enthalpy $\Delta H_{\text{rxn}}$ per mole of $\text{NH}_4\text{NO}_3$:
35 $\frac{\text{kJ}}{\text{mol}}$