QUESTION IMAGE
Question
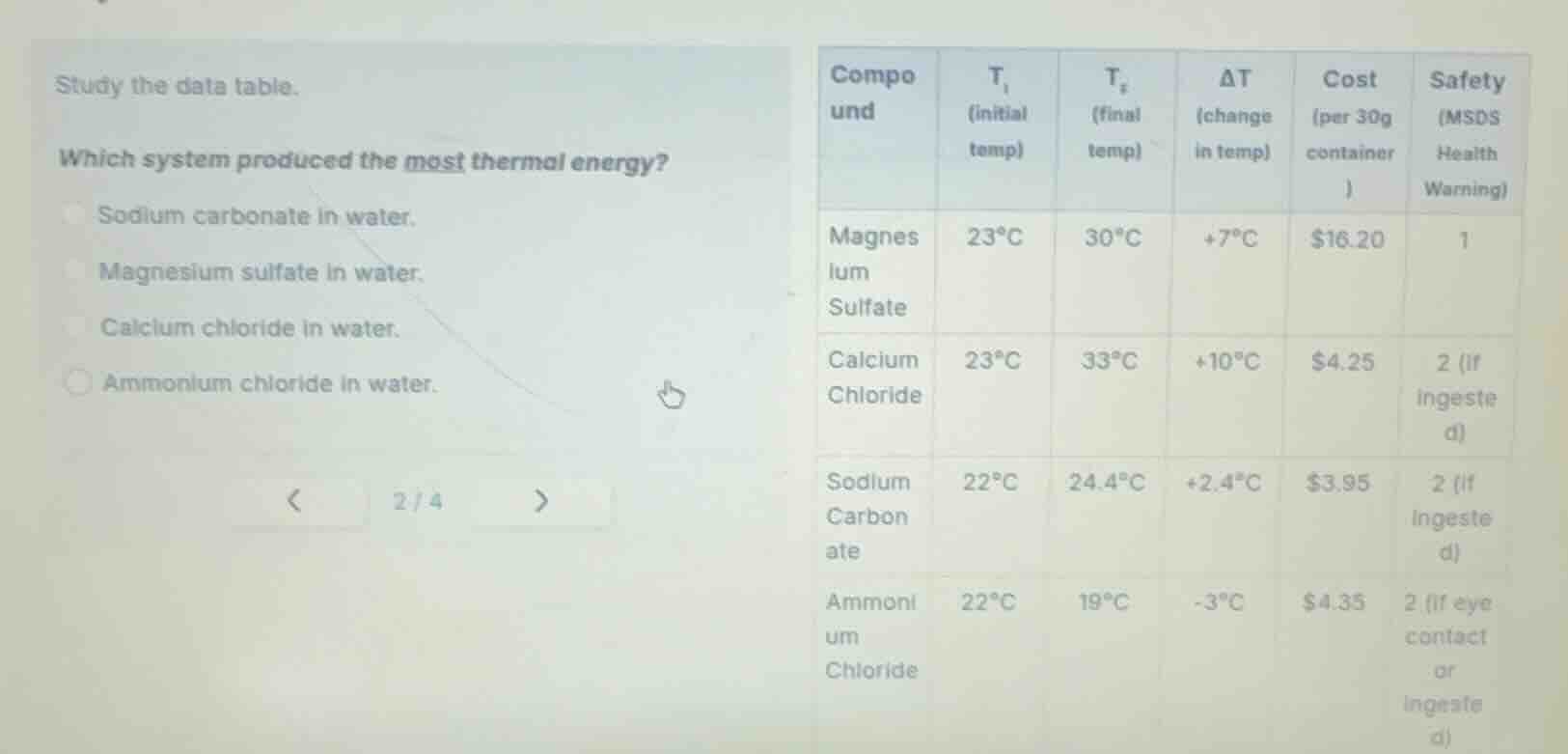
study the data table.
which system produced the most thermal energy?
sodium carbonate in water.
magnesium sulfate in water.
calcium chloride in water.
ammonium chloride in water.
| compound | $t_i$ (initial temp) | $t_f$ (final temp) | $delta t$ (change in temp) | cost (per 30g container) | safety (msds health warning) | ---- | ---- | ---- | ---- | ---- | ---- | magnesium sulfate | $23^circ c$ | $30^circ c$ | $+7^circ c$ | $16.20 | 1 | calcium chloride | $23^circ c$ | $33^circ c$ | $+10^circ c$ | $4.25 | 2 (if ingested) | sodium carbonate | $22^circ c$ | $24.4^circ c$ | $+2.4^circ c$ | $3.95 | 2 (if ingested) | ammonium chloride | $22^circ c$ | $19^circ c$ | $-3^circ c$ | $4.35 | 2 (if eye contact or ingested) |
Thermal energy released in a dissolution system is indicated by a positive temperature change (ΔT), where a larger ΔT means more thermal energy produced. Compare the ΔT values of each option: Magnesium sulfate (+7°C), Calcium chloride (+10°C), Sodium carbonate (+2.4°C), Ammonium chloride (-3°C, absorbs energy). Calcium chloride has the largest positive ΔT.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Calcium chloride in water.