QUESTION IMAGE
Question
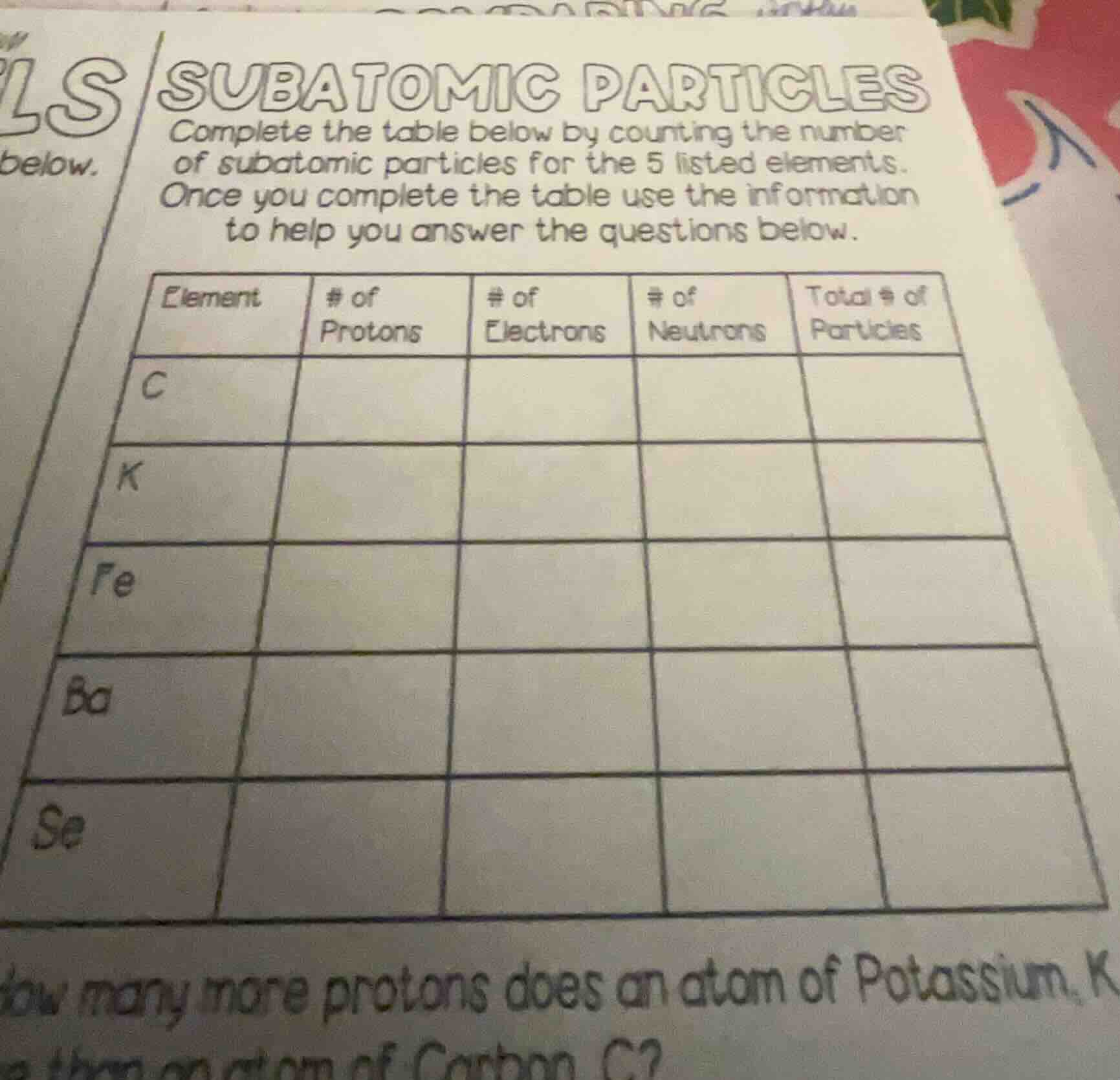
subatomic particles
complete the table below by counting the number
of subatomic particles for the 5 listed elements.
once you complete the table use the information
to help you answer the questions below.
element | # of protons | # of electrons | # of neutrons | total # of particles
c | | | |
k | | | |
fe | | | |
ba | | | |
se | | | |
how many more protons does an atom of potassium, k... than an atom of carbon, c?
Step1: Find proton count for each element
Protons = atomic number (from periodic table):
- C: $6$
- K: $19$
- Fe: $26$
- Ba: $56$
- Se: $34$
Step2: Find electron count for each element
Neutral atoms have electrons = protons:
- C: $6$
- K: $19$
- Fe: $26$
- Ba: $56$
- Se: $34$
Step3: Find neutron count for each element
Neutrons = mass number (most common isotope) - protons:
- C: $12 - 6 = 6$
- K: $39 - 19 = 20$
- Fe: $56 - 26 = 30$
- Ba: $137 - 56 = 81$
- Se: $79 - 34 = 45$
Step4: Calculate total particles per element
Total = protons + electrons + neutrons:
- C: $6 + 6 + 6 = 18$
- K: $19 + 19 + 20 = 58$
- Fe: $26 + 26 + 30 = 82$
- Ba: $56 + 56 + 81 = 193$
- Se: $34 + 34 + 45 = 113$
Step5: Answer the proton difference question
Subtract C protons from K protons:
$19 - 6 = 13$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Completed Table:
| Element | # of Protons | # of Electrons | # of Neutrons | Total # of Particles |
|---|---|---|---|---|
| K | 19 | 19 | 20 | 58 |
| Fe | 26 | 26 | 30 | 82 |
| Ba | 56 | 56 | 81 | 193 |
| Se | 34 | 34 | 45 | 113 |
Question Answer:
Potassium has 13 more protons than Carbon.