QUESTION IMAGE
Question
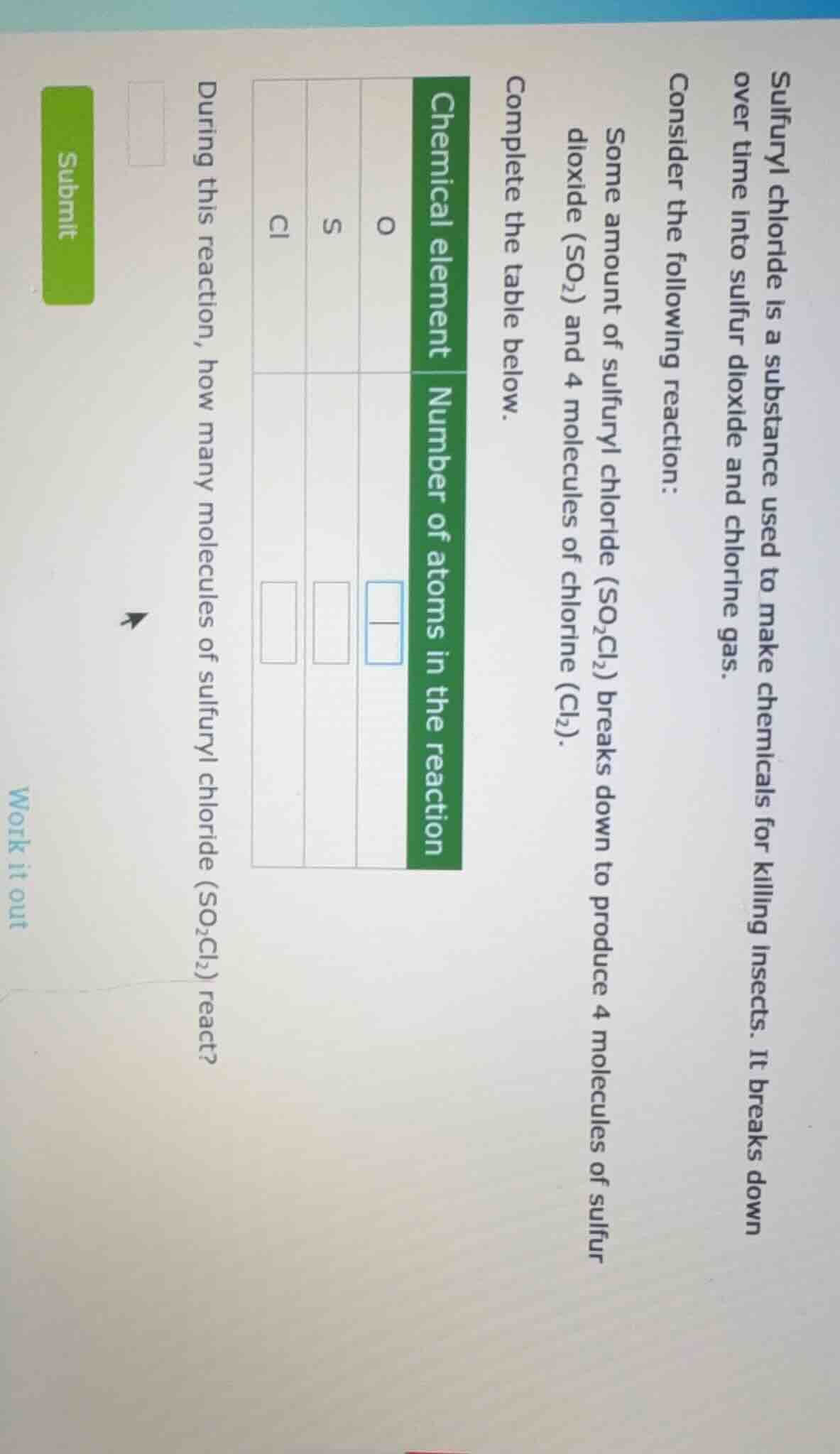
sulfuryl chloride is a substance used to make chemicals for killing insects. it breaks down over time into sulfur dioxide and chlorine gas. consider the following reaction: some amount of sulfuryl chloride (so₂cl₂) breaks down to produce 4 molecules of sulfur dioxide (so₂) and 4 molecules of chlorine (cl₂). complete the table below. chemical element | number of atoms in the reaction o | s | cl during this reaction, how many molecules of sulfuryl chloride (so₂cl₂) react? submit work it out
Step1: Determine the reaction stoichiometry
The reaction is the decomposition of sulfuryl chloride (\(SO_2Cl_2\)) into sulfur dioxide (\(SO_2\)) and chlorine gas (\(Cl_2\)). The balanced chemical equation is:
From the problem, 4 molecules of \(SO_2\) and 4 molecules of \(Cl_2\) are produced.
Step2: Relate moles of products to reactant
From the balanced equation, 1 mole (or molecule) of \(SO_2Cl_2\) produces 1 molecule of \(SO_2\) and 1 molecule of \(Cl_2\).
If 4 molecules of \(SO_2\) are produced, the number of \(SO_2Cl_2\) molecules that reacted is equal to the number of \(SO_2\) molecules produced (since the mole ratio of \(SO_2Cl_2\) to \(SO_2\) is \(1:1\)).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
4 molecules of sulfuryl chloride (\(SO_2Cl_2\)) react.