QUESTION IMAGE
Question
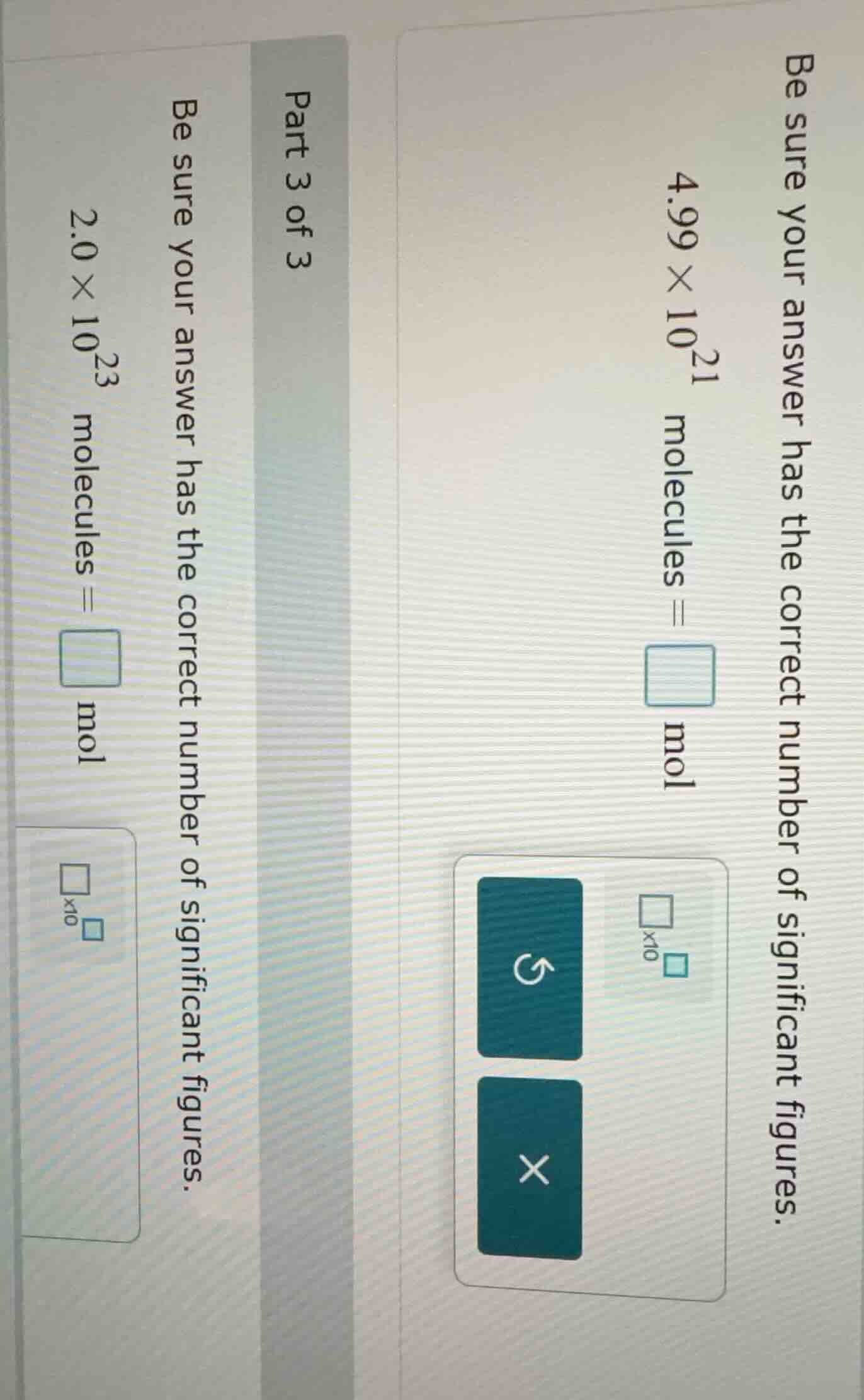
be sure your answer has the correct number of significant figures. \\(4.99 \times 10^{21}\\) molecules = \\(\square\\) mol \\(\square\times 10^{\square}\\) part 3 of 3 be sure your answer has the correct number of significant figures. \\(2.0 \times 10^{23}\\) molecules = \\(\square\\) mol \\(\square\times 10^{\square}\\)
First Sub - Question (for \(4.99\times10^{21}\) molecules to moles)
Step 1: Recall the formula for moles from molecules
The formula to convert the number of molecules to moles is \(n=\frac{N}{N_A}\), where \(n\) is the number of moles, \(N\) is the number of molecules, and \(N_A = 6.022\times 10^{23}\space\text{molecules/mol}\) (Avogadro's number).
Step 2: Substitute the values into the formula
We have \(N = 4.99\times 10^{21}\) molecules and \(N_A=6.022\times 10^{23}\space\text{molecules/mol}\). So \(n=\frac{4.99\times 10^{21}}{6.022\times 10^{23}}\).
Step 3: Perform the calculation
First, divide the coefficients: \(\frac{4.99}{6.022}\approx0.8286\). Then, divide the powers of 10: \(\frac{10^{21}}{10^{23}}=10^{- 2}\). Multiply these two results: \(0.8286\times10^{-2}=8.29\times 10^{-3}\space\text{mol}\) (rounded to three significant figures as the given value \(4.99\times 10^{21}\) has three significant figures).
Second Sub - Question (for \(2.0\times10^{23}\) molecules to moles)
Step 1: Recall the formula for moles from molecules
Again, use the formula \(n = \frac{N}{N_A}\), where \(N = 2.0\times 10^{23}\) molecules and \(N_A=6.022\times 10^{23}\space\text{molecules/mol}\).
Step 2: Substitute the values into the formula
\(n=\frac{2.0\times 10^{23}}{6.022\times 10^{23}}\).
Step 3: Perform the calculation
The \(10^{23}\) terms cancel out, and \(\frac{2.0}{6.022}\approx0.33\space\text{mol}\) (rounded to two significant figures as the given value \(2.0\times 10^{23}\) has two significant figures).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
For \(4.99\times10^{21}\) molecules: \(8.29\times 10^{-3}\space\text{mol}\)
For \(2.0\times10^{23}\) molecules: \(0.33\space\text{mol}\)