QUESTION IMAGE
Question
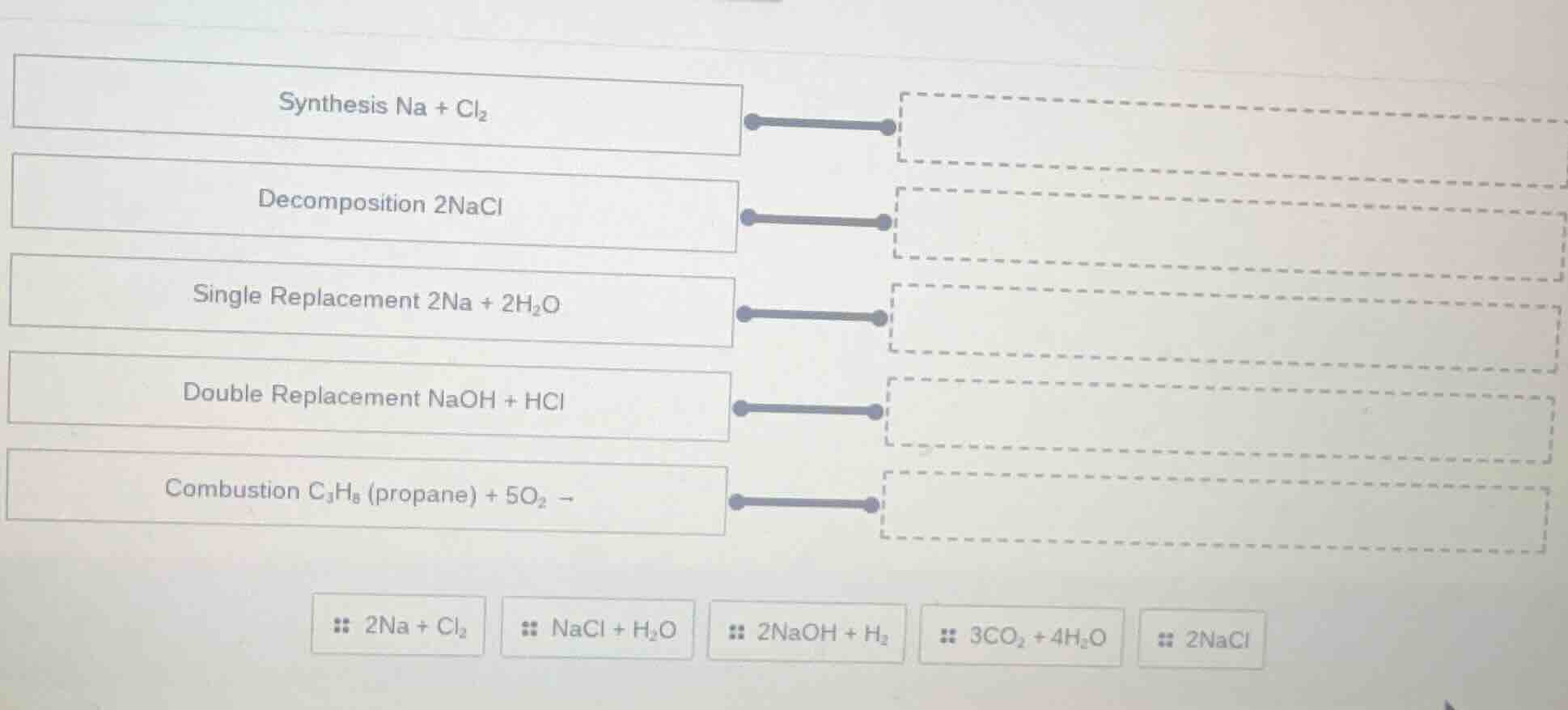
synthesis na + cl₂
decomposition 2nacl
single replacement 2na + 2h₂o
double replacement naoh + hcl
combustion c₃h₈ (propane) + 5o₂ →
:: 2na + cl₂
:: nacl + h₂o
:: 2naoh + h₂
:: 3co₂ + 4h₂o
:: 2nacl
Step1: Balance synthesis reaction
Synthesis: $\ce{Na + Cl2 -> 2NaCl}$
Step2: Balance decomposition reaction
Decomposition: $\ce{2NaCl -> 2Na + Cl2}$
Step3: Balance single replacement reaction
Single Replacement: $\ce{2Na + 2H2O -> 2NaOH + H2}$
Step4: Balance double replacement reaction
Double Replacement: $\ce{NaOH + HCl -> NaCl + H2O}$
Step5: Balance combustion reaction
Combustion: $\ce{C3H8 + 5O2 -> 3CO2 + 4H2O}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Synthesis $\ce{Na + Cl2}$ → $\boldsymbol{2NaCl}$
- Decomposition $\ce{2NaCl}$ → $\boldsymbol{2Na + Cl2}$
- Single Replacement $\ce{2Na + 2H2O}$ → $\boldsymbol{2NaOH + H2}$
- Double Replacement $\ce{NaOH + HCl}$ → $\boldsymbol{NaCl + H2O}$
- Combustion $\ce{C3H8 (propane) + 5O2}$ → $\boldsymbol{3CO2 + 4H2O}$