QUESTION IMAGE
Question
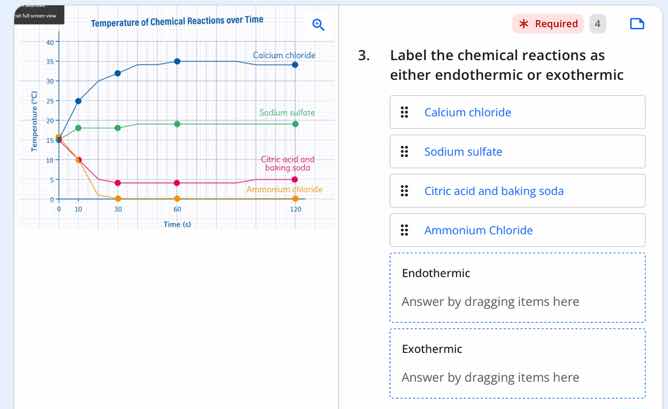
temperature of chemical reactions over time
- label the chemical reactions as either endothermic or exothermic
calcium chloride
sodium sulfate
citric acid and baking soda
ammonium chloride
endothermic
answer by dragging items here
exothermic
answer by dragging items here
Brief Explanations
- Calcium chloride: The graph shows its temperature rises from ~21°C to ~35°C, meaning it releases heat.
- Sodium sulfate: The graph shows its temperature rises slightly from ~21°C to ~22°C, meaning it releases heat.
- Citric acid and baking soda: The graph shows its temperature drops from ~21°C to ~8°C, meaning it absorbs heat.
- Ammonium chloride: The graph shows its temperature drops from ~21°C to ~5°C, meaning it absorbs heat.
Endothermic reactions absorb heat (temperature decreases), while exothermic reactions release heat (temperature increases).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
Endothermic
- Citric acid and baking soda
- Ammonium Chloride
Exothermic
- Calcium chloride
- Sodium sulfate