QUESTION IMAGE
Question
3.5.2 test (cs1): computer-scored unit test
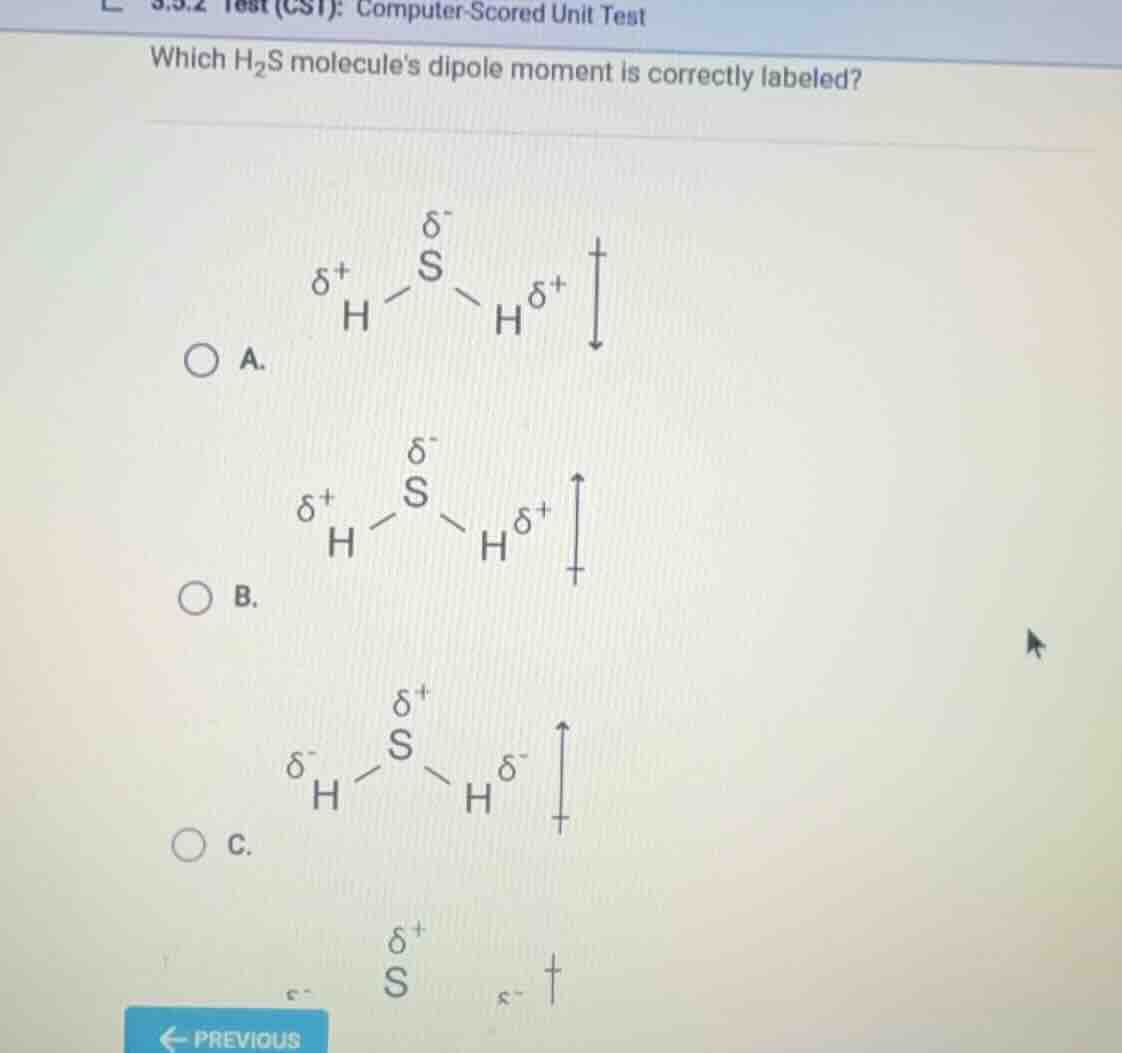
which h₂s molecules dipole moment is correctly labeled?
a.
δ⁻
s
δ⁺ h h δ⁺
↓
b.
δ⁻
s
δ⁺ h h δ⁺
↑
c.
δ⁺
s
δ⁻ h h δ⁻
↑
← previous
To determine the correct dipole moment for \( \ce{H2S} \), we analyze electronegativity: Sulfur (S) is more electronegative than Hydrogen (H), so S has a partial negative charge (\( \delta^- \)) and H has a partial positive charge (\( \delta^+ \)). The dipole moment vector points from the positive (H) to the negative (S) region. In option B, the dipole moment arrow points toward the more electronegative S (consistent with \( \delta^- \) on S and \( \delta^+ \) on H), while option A’s arrow direction is incorrect, and option C mislabels the charges.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
B. \( \boldsymbol{\delta^+}\ce{H - \overset{\delta^-}{S} - H^{\delta^+}} \) with the dipole moment arrow pointing upward (toward S)