QUESTION IMAGE
Question
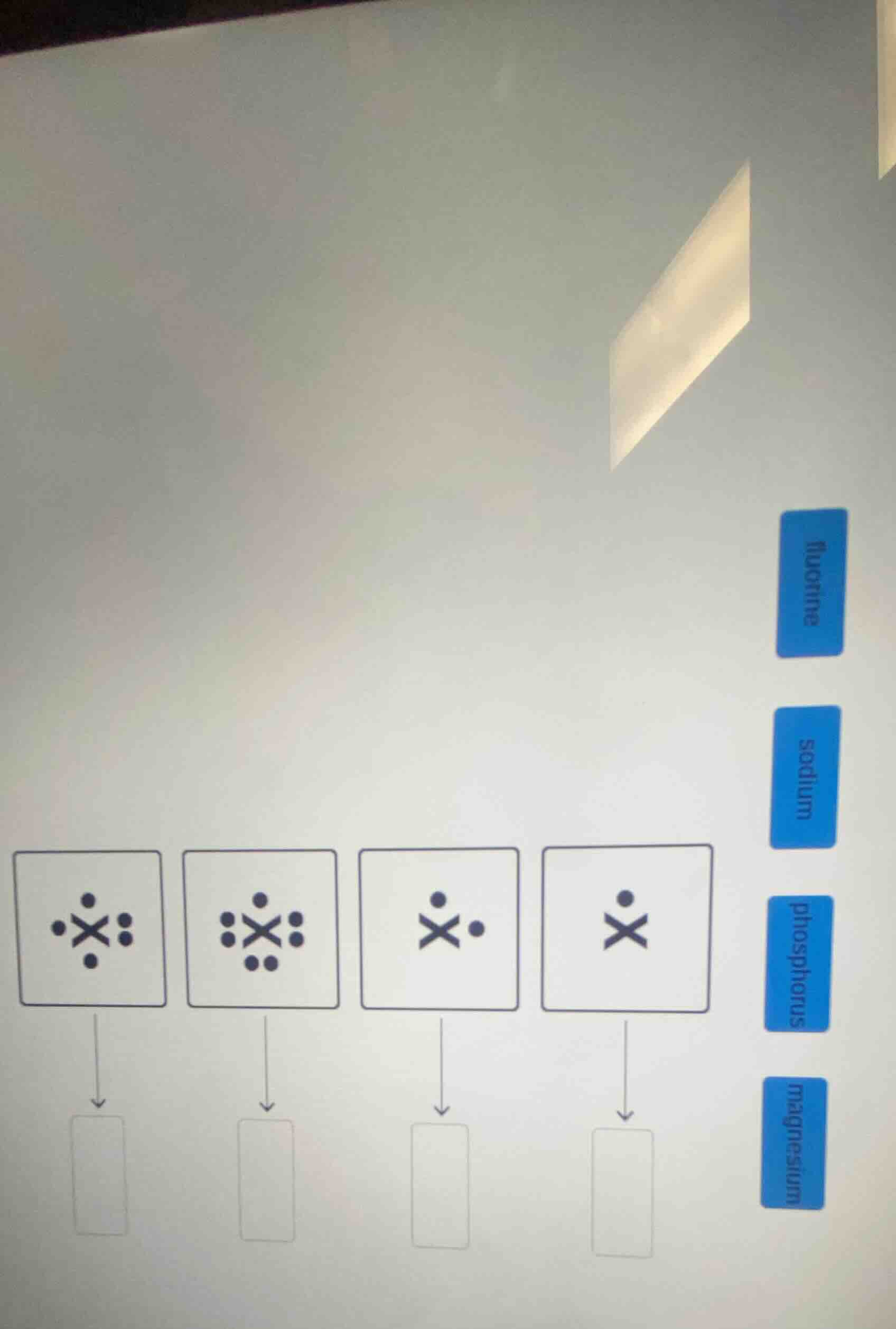
there are four squares with x and dots, and blue labels on the right: fluorine, sodium, phosphorus, magnesium. each square has an arrow to a blank rectangle.
To solve this problem, we analyze the pattern of dots around the "X" in each square and match it to the number of valence electrons of the elements (fluorine, sodium, phosphorus, magnesium) to determine the correct element for each square.
Step 1: Analyze the first square (4 dots around X)
The first square has 4 dots (2 on the left, 2 on the right, 1 top, 1 bottom? Wait, no—let's count the dots:
- First square: 4 dots (top, bottom, left, right—wait, no, looking at the image: first square has X with 4 dots (top, bottom, left (1), right (2)? Wait, no, let's re - examine. Wait, the first square: X with 4 dots (top, bottom, left (1), right (2)? No, maybe it's about valence electrons. Let's recall valence electrons:
- Fluorine (F): 7 valence electrons
- Sodium (Na): 1 valence electron
- Phosphorus (P): 5 valence electrons
- Magnesium (Mg): 2 valence electrons
Wait, maybe the dots represent valence electrons. Let's check the number of dots in each square:
- First square: Let's count the dots. Looking at the first square (leftmost), the X has 4 dots? Wait, no, the first square: top dot, bottom dot, left dot, right - two dots? Wait, maybe I miscounted. Wait, the first square: X with 4 dots (top, bottom, left (1), right (2)? No, maybe the number of dots is the number of valence electrons. Let's list valence electrons:
- Phosphorus (P): 5 valence electrons
- Magnesium (Mg): 2 valence electrons
- Sodium (Na): 1 valence electron
- Fluorine (F): 7 valence electrons
Wait, the squares have a decreasing number of dots. Let's count the dots in each square:
- Square 1 (leftmost): Let's count the dots around X. Top: 1, bottom: 1, left: 1, right: 2? No, that's 5. Oh! Square 1 has 5 dots. So that's phosphorus (P) with 5 valence electrons.
- Square 2: More dots. Let's count: top:1, bottom:2, left:2, right:2. Wait, that's 7 dots. So that's fluorine (F) with 7 valence electrons.
- Square 3: 2 dots (top:1, right:1). So that's magnesium (Mg) with 2 valence electrons.
- Square 4: 1 dot (top). So that's sodium (Na) with 1 valence electron.
Step 2: Match the squares to elements based on valence electrons
- Square 1 (5 dots): Phosphorus (P) has 5 valence electrons. So this square corresponds to phosphorus.
- Square 2 (7 dots): Fluorine (F) has 7 valence electrons. So this square corresponds to fluorine.
- Square 3 (2 dots): Magnesium (Mg) has 2 valence electrons. So this square corresponds to magnesium.
- Square 4 (1 dot): Sodium (Na) has 1 valence electron. So this square corresponds to sodium.
Final Matching
- First square (leftmost) with 5 dots: Phosphorus
- Second square with 7 dots: Fluorine
- Third square with 2 dots: Magnesium
- Fourth square with 1 dot: Sodium
So the answers (from left to right for the empty boxes below the squares) are:
- Phosphorus
- Fluorine
- Magnesium
- Sodium
(Note: The above solution assumes that the dots around the "X" represent the valence electrons of the element, and we match the number of dots to the number of valence electrons of each element (fluorine, sodium, phosphorus, magnesium) to determine the correct element for each square.)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve this problem, we analyze the pattern of dots around the "X" in each square and match it to the number of valence electrons of the elements (fluorine, sodium, phosphorus, magnesium) to determine the correct element for each square.
Step 1: Analyze the first square (4 dots around X)
The first square has 4 dots (2 on the left, 2 on the right, 1 top, 1 bottom? Wait, no—let's count the dots:
- First square: 4 dots (top, bottom, left, right—wait, no, looking at the image: first square has X with 4 dots (top, bottom, left (1), right (2)? Wait, no, let's re - examine. Wait, the first square: X with 4 dots (top, bottom, left (1), right (2)? No, maybe it's about valence electrons. Let's recall valence electrons:
- Fluorine (F): 7 valence electrons
- Sodium (Na): 1 valence electron
- Phosphorus (P): 5 valence electrons
- Magnesium (Mg): 2 valence electrons
Wait, maybe the dots represent valence electrons. Let's check the number of dots in each square:
- First square: Let's count the dots. Looking at the first square (leftmost), the X has 4 dots? Wait, no, the first square: top dot, bottom dot, left dot, right - two dots? Wait, maybe I miscounted. Wait, the first square: X with 4 dots (top, bottom, left (1), right (2)? No, maybe the number of dots is the number of valence electrons. Let's list valence electrons:
- Phosphorus (P): 5 valence electrons
- Magnesium (Mg): 2 valence electrons
- Sodium (Na): 1 valence electron
- Fluorine (F): 7 valence electrons
Wait, the squares have a decreasing number of dots. Let's count the dots in each square:
- Square 1 (leftmost): Let's count the dots around X. Top: 1, bottom: 1, left: 1, right: 2? No, that's 5. Oh! Square 1 has 5 dots. So that's phosphorus (P) with 5 valence electrons.
- Square 2: More dots. Let's count: top:1, bottom:2, left:2, right:2. Wait, that's 7 dots. So that's fluorine (F) with 7 valence electrons.
- Square 3: 2 dots (top:1, right:1). So that's magnesium (Mg) with 2 valence electrons.
- Square 4: 1 dot (top). So that's sodium (Na) with 1 valence electron.
Step 2: Match the squares to elements based on valence electrons
- Square 1 (5 dots): Phosphorus (P) has 5 valence electrons. So this square corresponds to phosphorus.
- Square 2 (7 dots): Fluorine (F) has 7 valence electrons. So this square corresponds to fluorine.
- Square 3 (2 dots): Magnesium (Mg) has 2 valence electrons. So this square corresponds to magnesium.
- Square 4 (1 dot): Sodium (Na) has 1 valence electron. So this square corresponds to sodium.
Final Matching
- First square (leftmost) with 5 dots: Phosphorus
- Second square with 7 dots: Fluorine
- Third square with 2 dots: Magnesium
- Fourth square with 1 dot: Sodium
So the answers (from left to right for the empty boxes below the squares) are:
- Phosphorus
- Fluorine
- Magnesium
- Sodium
(Note: The above solution assumes that the dots around the "X" represent the valence electrons of the element, and we match the number of dots to the number of valence electrons of each element (fluorine, sodium, phosphorus, magnesium) to determine the correct element for each square.)