QUESTION IMAGE
Question
there are three types of chemical bonds: covalent, ionic, and metallic.
a covalent bond is formed when two atoms share one or more pairs of electrons. covalent bonds tend to form between nonmetal atoms as their electronegativity difference is relatively small. a compound held together by covalent bonds is called a covalent compound.
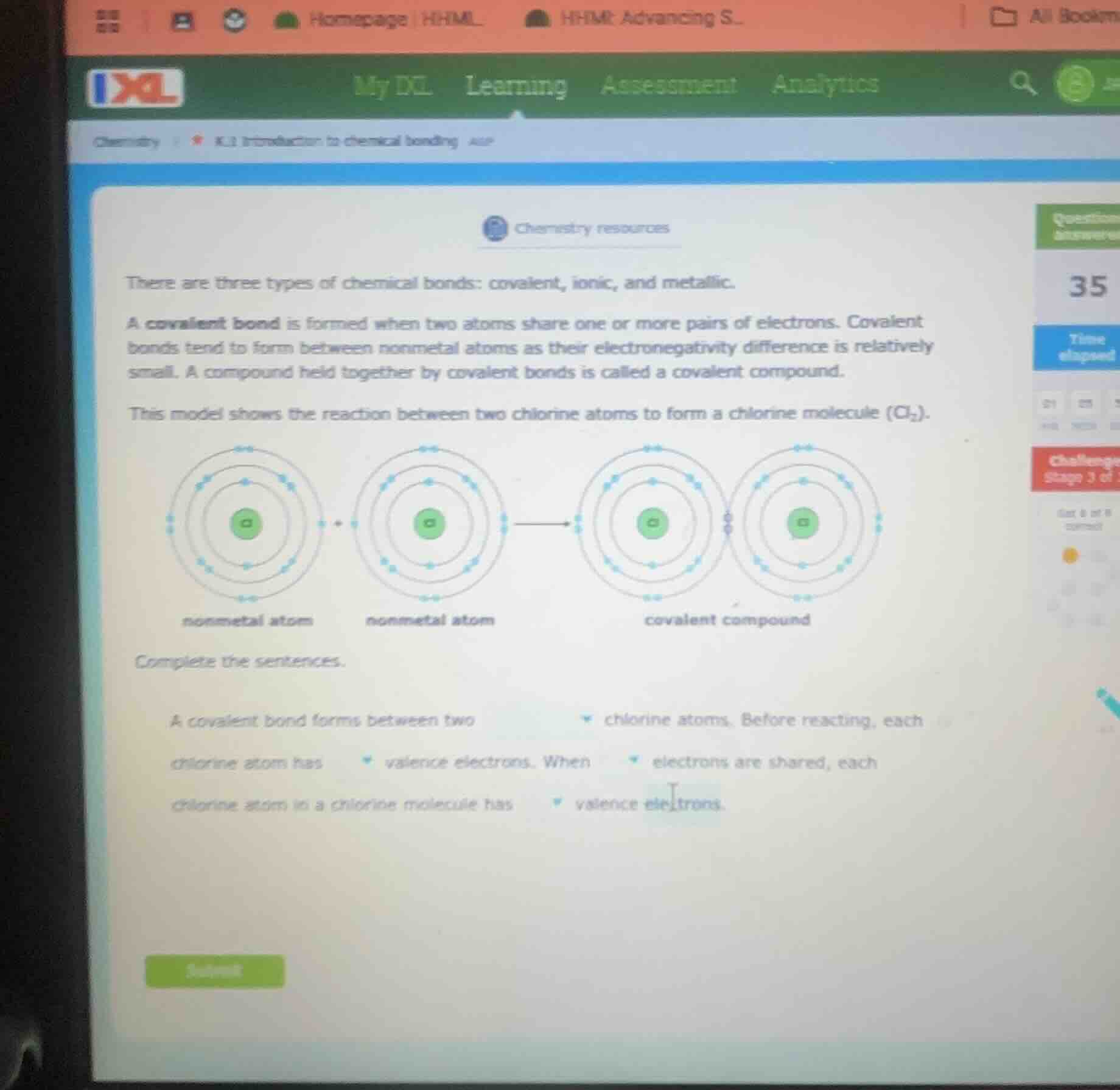
this model shows the reaction between two chlorine atoms to form a chlorine molecule ($cl_2$).
complete the sentences.
a covalent bond forms between two chlorine atoms. before reacting, each chlorine atom has valence electrons. when electrons are shared, each chlorine atom in a chlorine molecule has valence electrons.
Step1: Identify bond type context
Chlorine is a nonmetal, so the bond is between two nonmetal atoms.
Step2: Find chlorine's valence electrons
Chlorine (atomic number 17) has an electron configuration of $1s^22s^22p^63s^23p^5$, so valence electrons (3rd shell) = 7.
Step3: Determine shared electrons
To form a stable octet, each Cl shares 1 electron, so total shared electrons = 2 (1 pair).
Step4: Calculate valence electrons in molecule
Each Cl retains 6 unshared valence electrons + 1 shared electron from the other Cl, so total = 6 + 1 = 8.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A covalent bond forms between two nonmetal chlorine atoms. Before reacting, each chlorine atom has 7 valence electrons. When 2 electrons are shared, each chlorine atom in a chlorine molecule has 8 valence electrons.