QUESTION IMAGE
Question
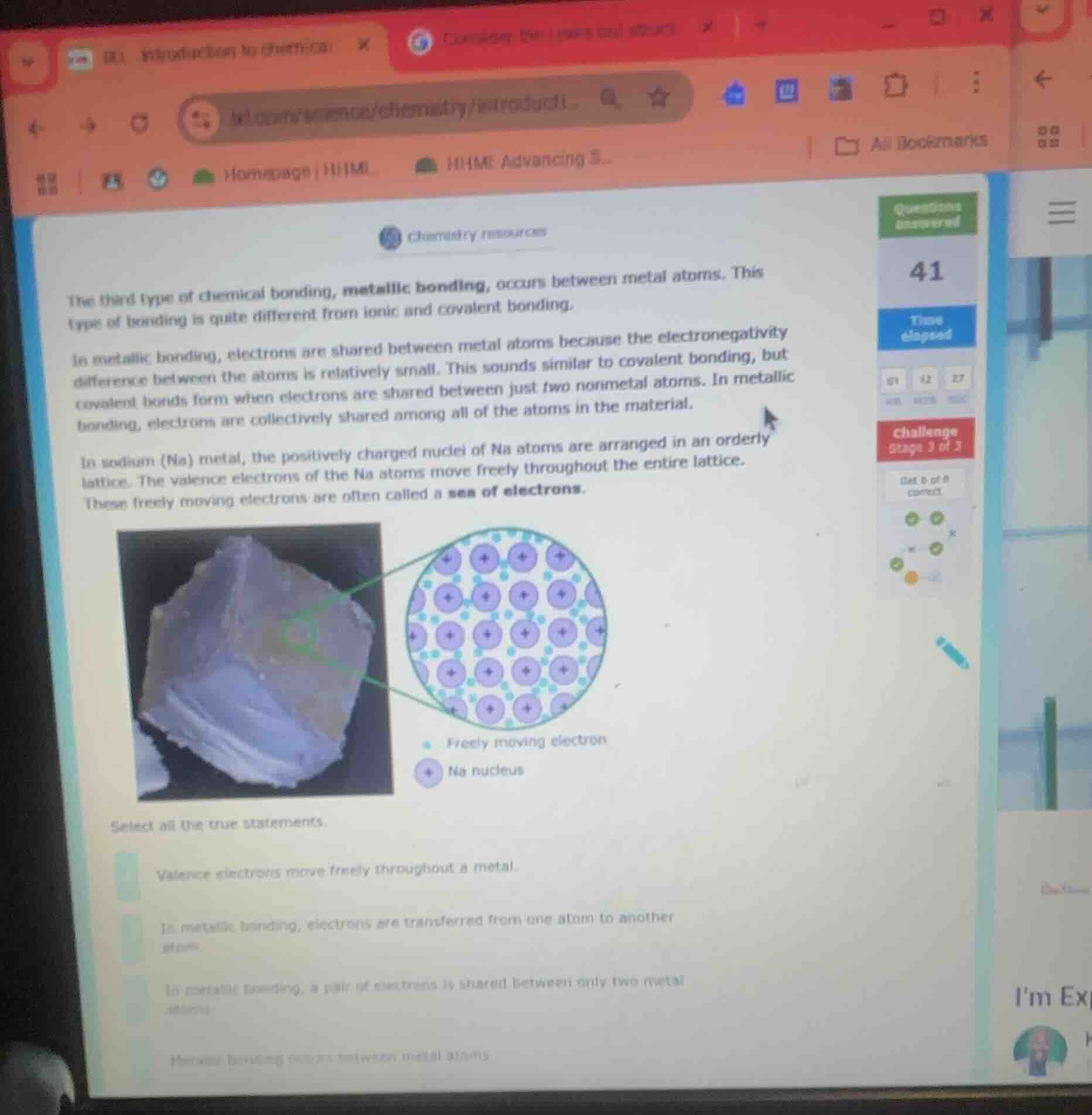
the third type of chemical bonding, metallic bonding, occurs between metal atoms. this type of bonding is quite different from ionic and covalent bonding.in metallic bonding, electrons are shared between metal atoms because the electronegativity difference between the atoms is relatively small. this sounds similar to covalent bonding, but covalent bonds form when electrons are shared between just two nonmetal atoms. in metallic bonding, electrons are collectively shared among all of the atoms in the material.in sodium (na) metal, the positively charged nuclei of na atoms are arranged in an orderly lattice. the valence electrons of the na atoms move freely throughout the entire lattice. these freely moving electrons are often called a sea of electrons.select all the true statements.valence electrons move freely throughout a metal.in metallic bonding, electrons are transferred from one atom to another atom.in metallic bonding, a pair of electrons is shared between only two metal atoms.metallic bonding occurs between metal atoms.
- Check the first statement: The text states "The valence electrons of the Na atoms move freely throughout the entire lattice" for sodium metal, which generalizes to valence electrons moving freely in any metal. This is true.
- Check the second statement: Electron transfer occurs in ionic bonding, not metallic bonding (metallic bonding uses collective electron sharing). This is false.
- Check the third statement: The text notes covalent bonds share electrons between two atoms, while metallic bonding shares electrons collectively among all atoms. This is false.
- Check the fourth statement: The text opens with "metallic bonding, occurs between metal atoms." This is true.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- Valence electrons move freely throughout a metal.
- Metallic bonding occurs between metal atoms.