QUESTION IMAGE
Question
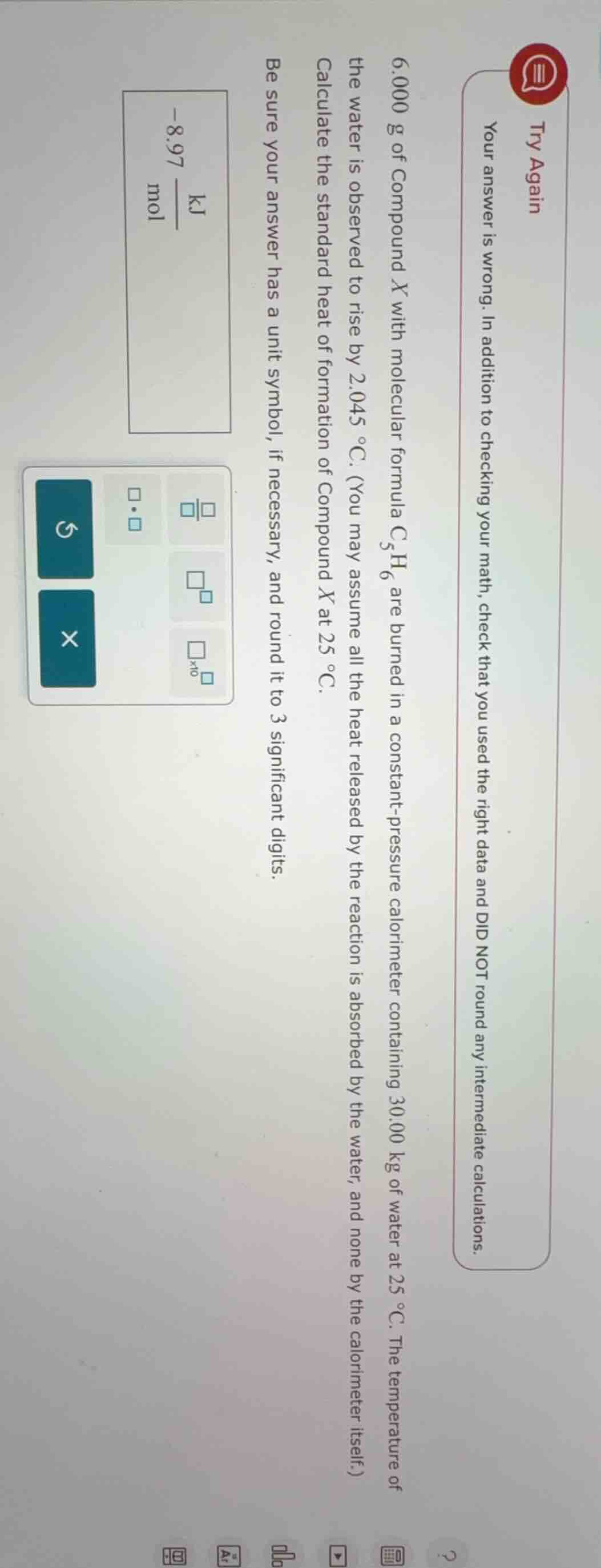
try again
your answer is wrong. in addition to checking your math, check that you used the right data and did not round any intermediate calculations.
6.000 g of compound $x$ with molecular formula $c_5h_6$ are burned in a constant-pressure calorimeter containing 30.00 kg of water at 25 °c. the temperature of the water is observed to rise by 2.045 °c. (you may assume all the heat released by the reaction is absorbed by the water, and none by the calorimeter itself.)
calculate the standard heat of formation of compound $x$ at 25 °c.
be sure your answer has a unit symbol, if necessary, and round it to 3 significant digits.
$-8.97\\ \frac{\text{kj}}{\text{mol}}$
Step1: Calculate heat absorbed by water
Use $q = mc\Delta T$, where $m=30.00\ \text{kg}=30000\ \text{g}$, $c=4.184\ \frac{\text{J}}{\text{g}^\circ\text{C}}$, $\Delta T=2.045^\circ\text{C}$
$q = 30000\ \text{g} \times 4.184\ \frac{\text{J}}{\text{g}^\circ\text{C}} \times 2.045^\circ\text{C} = 257540.4\ \text{J} = 257.5404\ \text{kJ}$
Step2: Find moles of Compound X
Molar mass of $\text{C}_5\text{H}_6$: $5\times12.01 + 6\times1.008 = 66.108\ \frac{\text{g}}{\text{mol}}$
Moles $n = \frac{6.000\ \text{g}}{66.108\ \frac{\text{g}}{\text{mol}}} = 0.09076\ \text{mol}$
Step3: Calculate heat of combustion
Heat released per mole: $\Delta H_{\text{comb}} = -\frac{q}{n} = -\frac{257.5404\ \text{kJ}}{0.09076\ \text{mol}} = -2837.6\ \frac{\text{kJ}}{\text{mol}}$
Step4: Write combustion reaction
$\text{C}_5\text{H}_6(l) + \frac{13}{2}\text{O}_2(g)
ightarrow 5\text{CO}_2(g) + 3\text{H}_2\text{O}(l)$
Step5: Use $\Delta H^\circ_{\text{rxn}} = \sum \Delta H^\circ_f(\text{products}) - \sum \Delta H^\circ_f(\text{reactants})$
Known values: $\Delta H^\circ_f(\text{CO}_2) = -393.5\ \frac{\text{kJ}}{\text{mol}}$, $\Delta H^\circ_f(\text{H}_2\text{O}) = -285.8\ \frac{\text{kJ}}{\text{mol}}$, $\Delta H^\circ_f(\text{O}_2)=0$
$-2837.6 = [5\times(-393.5) + 3\times(-285.8)] - \Delta H^\circ_f(\text{C}_5\text{H}_6)$
Step6: Solve for $\Delta H^\circ_f(\text{C}_5\text{H}_6)$
Calculate product sum: $5\times(-393.5) + 3\times(-285.8) = -1967.5 - 857.4 = -2824.9\ \frac{\text{kJ}}{\text{mol}}$
$\Delta H^\circ_f(\text{C}_5\text{H}_6) = -2824.9 + 2837.6 = 12.7\ \frac{\text{kJ}}{\text{mol}}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$12.7\ \frac{\text{kJ}}{\text{mol}}$