QUESTION IMAGE
Question
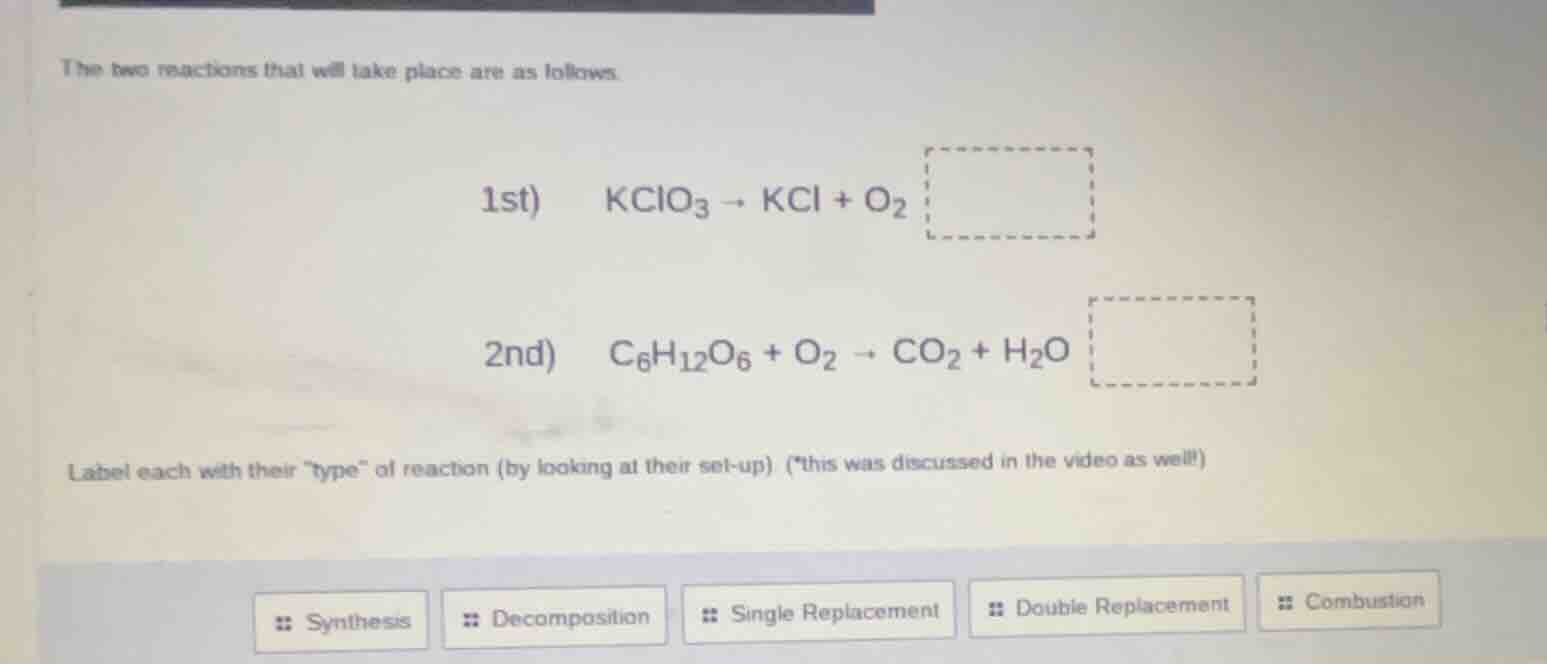
the two reactions that will take place are as follows.
1st) ( ce{kclo_3 -> kcl + o_2} )
2nd) ( ce{c_6h_{12}o_6 + o_2 -> co_2 + h_2o} )
label each with their \type\ of reaction (by looking at their set - up) (this was discussed in the video as well!)
options: synthesis, decomposition, single replacement, double replacement, combustion
For the first reaction ($\ce{KClO3 \to KCl + O2}$), a single compound breaks down into simpler substances (KCl and O₂), which fits the definition of a decomposition reaction. For the second reaction ($\ce{C6H12O6 + O2 \to CO2 + H2O}$), a hydrocarbon (glucose) reacts with oxygen to produce carbon dioxide and water, which is characteristic of a combustion reaction.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
1st reaction: Decomposition
2nd reaction: Combustion