QUESTION IMAGE
Question
types of reactions
quiz complete
time remaining
45:26
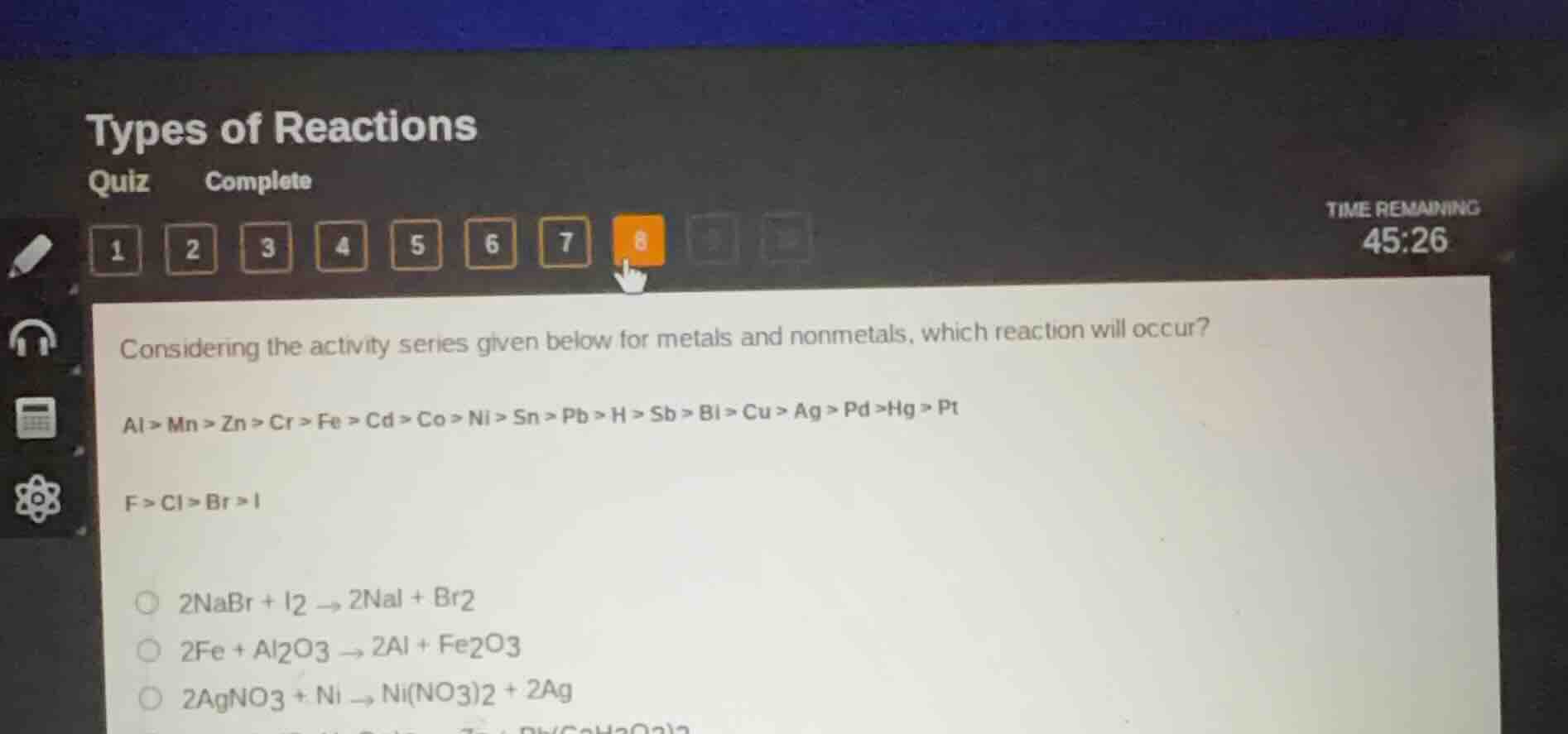
considering the activity series given below for metals and nonmetals, which reaction will occur?
al > mn > zn > cr > fe > cd > co > ni > sn > pb > h > sb > bi > cu > ag > pd >hg > pt
f > cl > br > i
$2nabr + i_2 \
ightarrow 2nai + br_2$
$2fe + al_2o_3 \
ightarrow 2al + fe_2o_3$
$2agno_3 + ni \
ightarrow ni(no_3)_2 + 2ag$
Step1: Recall displacement reaction rule
A more reactive element displaces a less reactive element from its compound.
Step2: Analyze nonmetal reaction option
Given nonmetal series: $\text{F} > \text{Cl} > \text{Br} > \text{I}$. For $\ce{2NaBr + I2 -> 2NaI + Br2}$, $\text{I}$ is less reactive than $\text{Br}$, so no reaction.
Step3: Analyze first metal reaction option
Given metal series: $\text{Al} > \text{Fe}$. For $\ce{2Fe + Al2O3 -> 2Al + Fe2O3}$, $\text{Fe}$ is less reactive than $\text{Al}$, so no reaction.
Step4: Analyze second metal reaction option
Given metal series: $\text{Ni} > \text{Ag}$. For $\ce{2AgNO3 + Ni -> Ni(NO3)2 + 2Ag}$, $\text{Ni}$ is more reactive than $\text{Ag}$, so reaction occurs.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\boldsymbol{2AgNO3 + Ni
ightarrow Ni(NO3)2 + 2Ag}$