QUESTION IMAGE
Question
types of reactions
quiz complete
1 2
time remaining
54:09
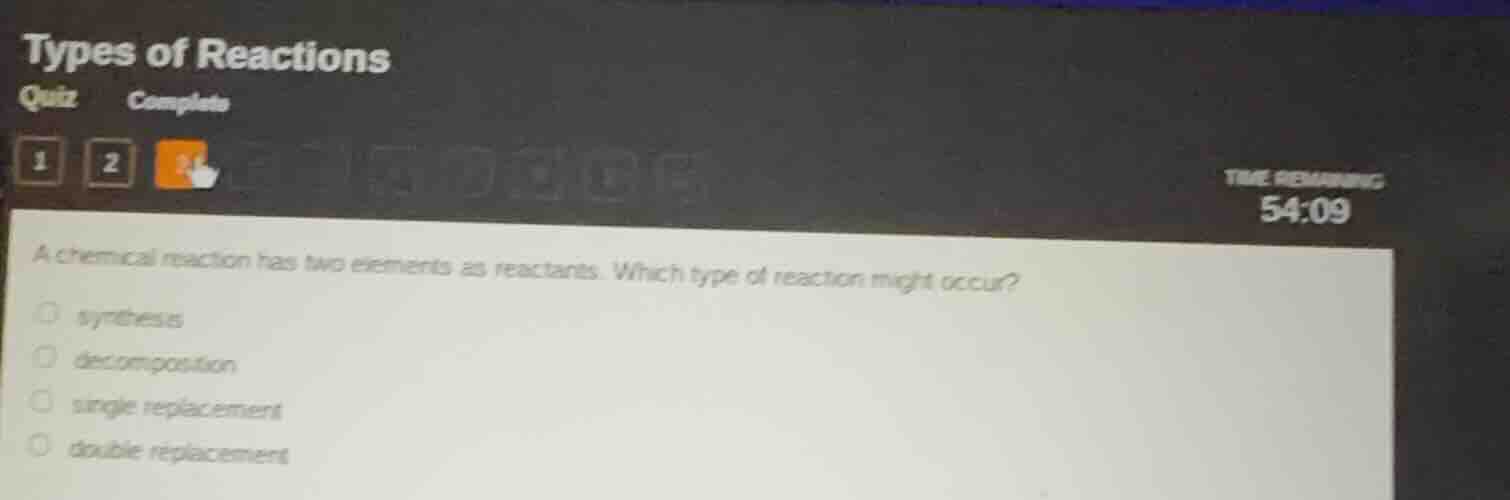
a chemical reaction has two elements as reactants. which type of reaction might occur?
synthesis
decomposition
single replacement
double replacement
Brief Explanations
- A synthesis reaction combines two or more simple substances (like elements) into a more complex product, fitting the reactant description.
- Decomposition reactions break down a single compound into simpler substances, so it cannot start with two elements.
- Single replacement reactions involve one element replacing another in a compound, requiring a compound as one reactant.
- Double replacement reactions swap ions between two compounds, so they need compounds as reactants, not elements.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
synthesis