QUESTION IMAGE
Question
types of reactions
quiz complete
time remaining
55:50
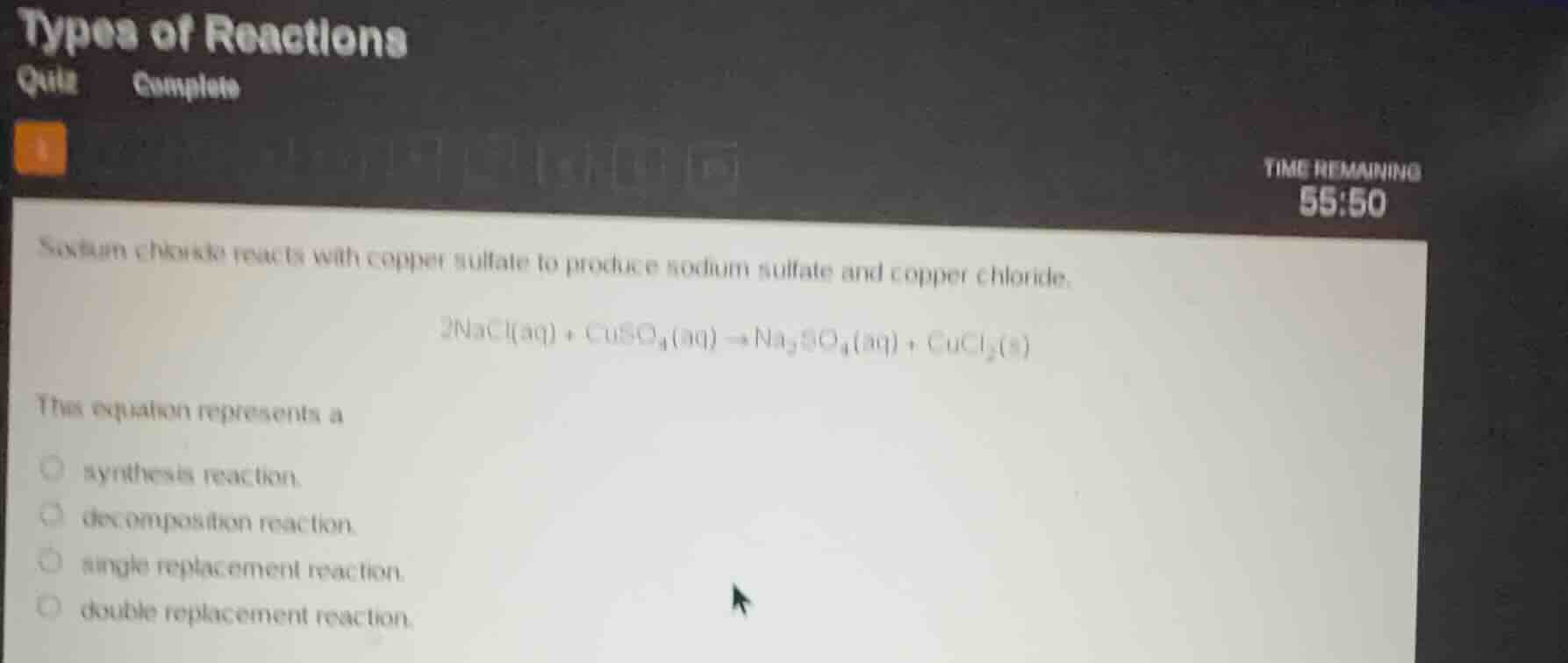
sodium chloride reacts with copper sulfate to produce sodium sulfate and copper chloride.
$2nacl(aq) + cuso_4(aq) \
ightarrow na_2so_4(aq) + cucl_2(s)$
this equation represents a
synthesis reaction.
decomposition reaction.
single replacement reaction.
double replacement reaction.
A double replacement reaction involves two ionic compounds swapping their positive and negative ions to form new compounds. In the given reaction, sodium ($\text{Na}^+$) and copper ($\text{Cu}^{2+}$) swap their anions (chloride $\text{Cl}^-$ and sulfate $\text{SO}_4^{2-}$) to form sodium sulfate and copper chloride, which matches this reaction type. Synthesis reactions combine reactants into one product, decomposition breaks one reactant into multiple products, and single replacement involves one element replacing another in a compound, none of which fit here.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
double replacement reaction.