QUESTION IMAGE
Question
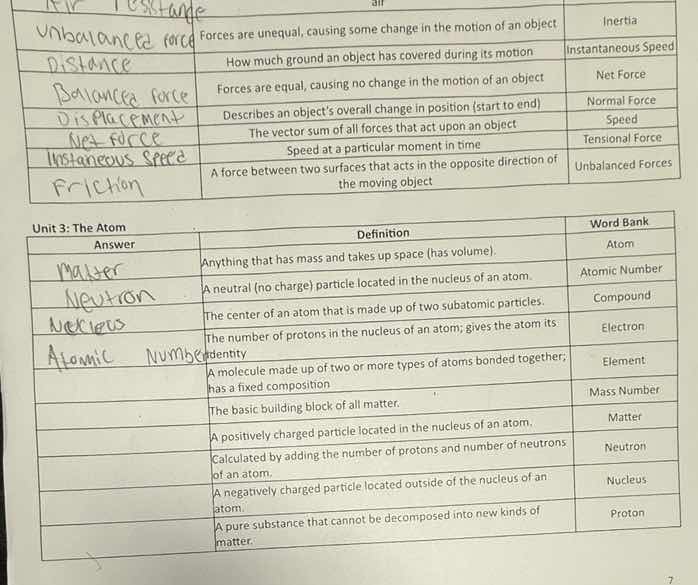
unbalanced force: forces are unequal, causing some change in the motion of an object. inertia
distance: how much ground an object has covered during its motion. instantaneous speed
balanced force: forces are equal, causing no change in the motion of an object. net force
displacement: describes an objects overall change in position (start to end). normal force
net force: the vector sum of all forces that act upon an object. speed
instantaneous speed: speed at a particular moment in time. tensional force
friction: a force between two surfaces that acts in the opposite direction of the moving object. unbalanced forces
unit 3: the atom
answer\tdefinition\tword bank
matter\tanything that has mass and takes up space (has volume).\tatom
neutron\ta neutral (no charge) particle located in the nucleus of an atom.\tatomic number
nucleus\tthe center of an atom that is made up of two subatomic particles.\tcompound
atomic number\tthe number of protons in the nucleus of an atom; gives the atom its identity.\telectron
\ta molecule made up of two or more types of atoms bonded together; has a fixed composition.\telement
\tthe basic building block of all matter.\tmass number
\ta positively charged particle located in the nucleus of an atom.\tmatter
\tcalculated by adding the number of protons and number of neutrons of an atom.\tneutron
\ta negatively charged particle located outside of the nucleus of an atom.\tnucleus
\ta pure substance that cannot be decomposed into new kinds of matter.\tproton
To solve this matching problem for Unit 3: The Atom, we analyze each definition and match it with the correct term from the word bank:
1. "Anything that has mass and takes up space (has volume)."
This defines Matter.
2. "A neutral (no charge) particle located in the nucleus of an atom."
This describes a Neutron.
3. "The center of an atom that is made up of two subatomic particles."
This refers to the Nucleus (contains protons and neutrons).
4. "The number of protons in the nucleus of an atom; gives the atom its identity."
This is the Atomic Number.
5. "A molecule made up of two or more types of atoms bonded together; has a fixed composition."
This defines a Compound.
6. "The basic building block of all matter."
This is an Atom.
7. "A positively charged particle located in the nucleus of an atom."
This describes a Proton.
8. "Calculated by adding the number of protons and number of neutrons of an atom."
This is the Mass Number.
9. "A negatively charged particle located outside of the nucleus of an atom."
This refers to an Electron.
10. "A pure substance that cannot be decomposed into new kinds of matter."
This defines an Element.
Final Matches (Answer Column):
- Matter
- Neutron
- Nucleus
- Atomic Number
- Compound
- Atom
- Proton
- Mass Number
- Electron
- Element
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
To solve this matching problem for Unit 3: The Atom, we analyze each definition and match it with the correct term from the word bank:
1. "Anything that has mass and takes up space (has volume)."
This defines Matter.
2. "A neutral (no charge) particle located in the nucleus of an atom."
This describes a Neutron.
3. "The center of an atom that is made up of two subatomic particles."
This refers to the Nucleus (contains protons and neutrons).
4. "The number of protons in the nucleus of an atom; gives the atom its identity."
This is the Atomic Number.
5. "A molecule made up of two or more types of atoms bonded together; has a fixed composition."
This defines a Compound.
6. "The basic building block of all matter."
This is an Atom.
7. "A positively charged particle located in the nucleus of an atom."
This describes a Proton.
8. "Calculated by adding the number of protons and number of neutrons of an atom."
This is the Mass Number.
9. "A negatively charged particle located outside of the nucleus of an atom."
This refers to an Electron.
10. "A pure substance that cannot be decomposed into new kinds of matter."
This defines an Element.
Final Matches (Answer Column):
- Matter
- Neutron
- Nucleus
- Atomic Number
- Compound
- Atom
- Proton
- Mass Number
- Electron
- Element