QUESTION IMAGE
Question
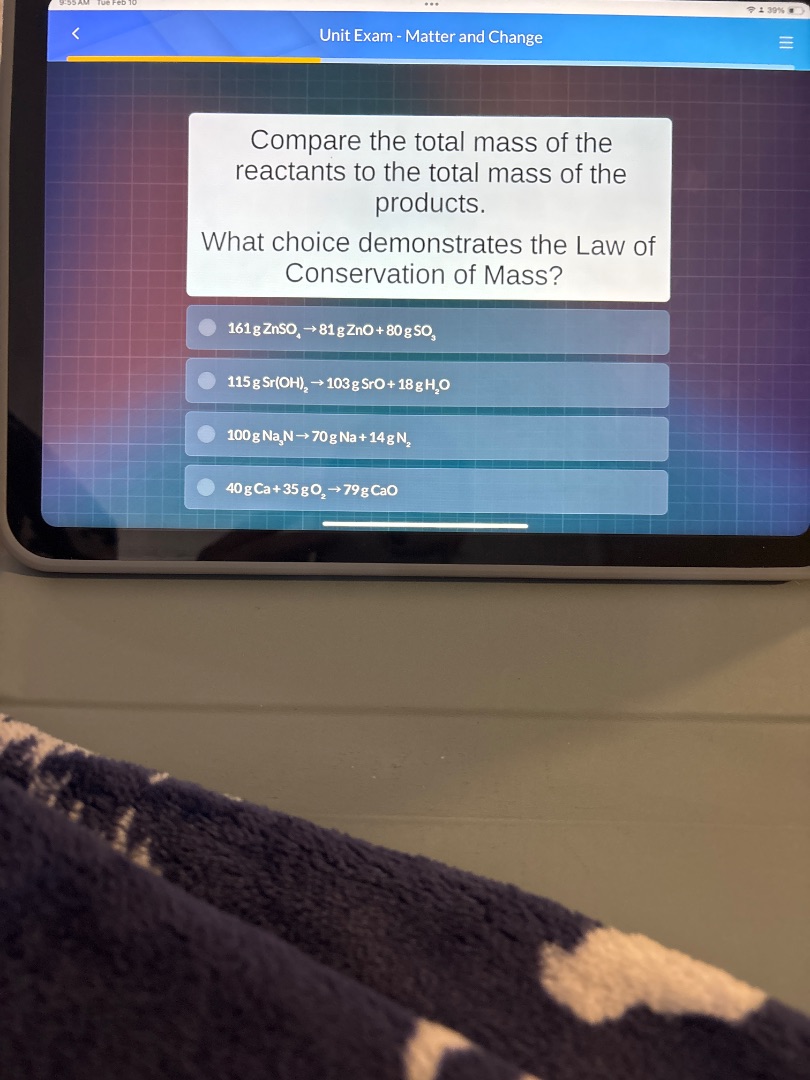
unit exam - matter and change
compare the total mass of the reactants to the total mass of the products.
what choice demonstrates the law of conservation of mass?
161 g znso₄ → 81 g zno + 80 g so₃
115 g sr(oh)₂ → 103 g sro + 18 g h₂o
100 g na₃n → 70 g na + 14 g n₂
40 g ca + 35 g o₂ → 79 g cao
Step1: Calculate reactant vs product mass
For first option: $161\ \text{g}$ vs $81+80=161\ \text{g}$
Step2: Calculate reactant vs product mass
For second option: $115\ \text{g}$ vs $103+18=121\ \text{g}$
Step3: Calculate reactant vs product mass
For third option: $100\ \text{g}$ vs $70+14=84\ \text{g}$
Step4: Calculate reactant vs product mass
For fourth option: $40+35=75\ \text{g}$ vs $79\ \text{g}$
Step5: Match to Law of Conservation
Only first option has equal masses.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
161 g ZnSO₄ → 81 g ZnO + 80 g SO₃