QUESTION IMAGE
Question
unit 7 quiz: 7.1-7.2
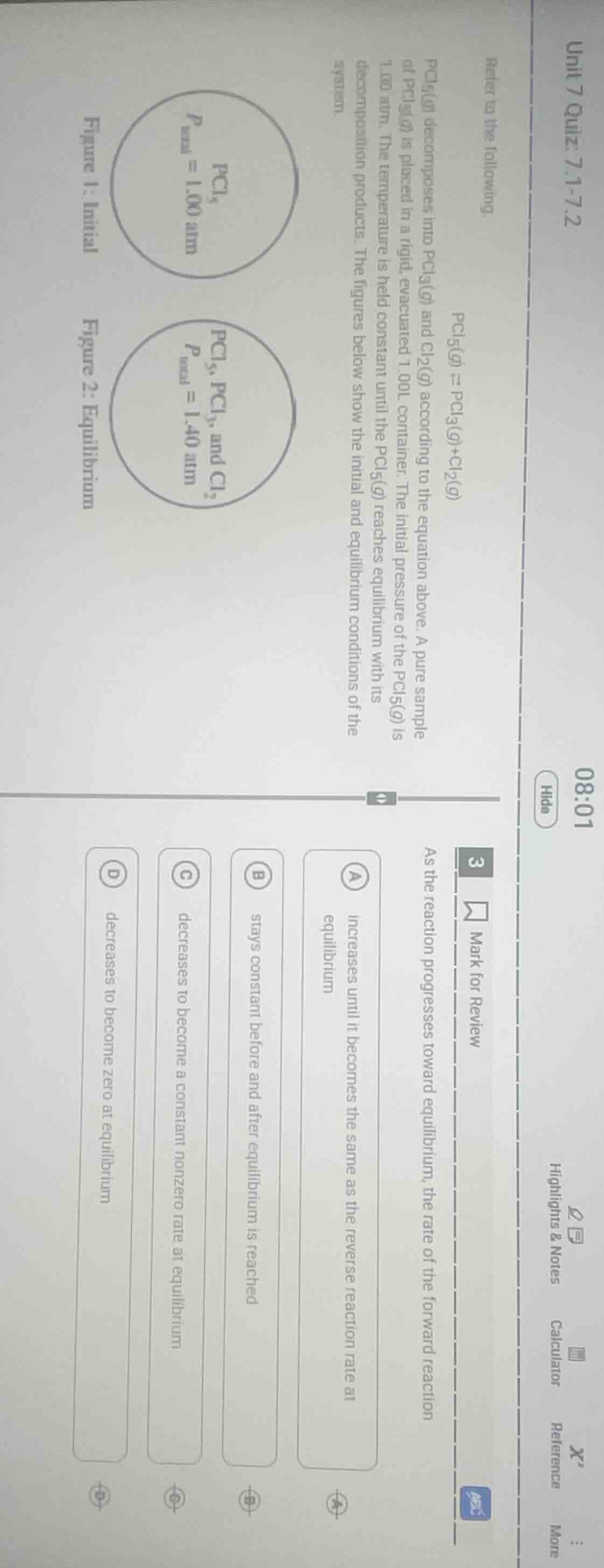
refer to the following
pcl₅(g) ⇌ pcl₃(g)+cl₂(g)
pcl₅(g) decomposes into pcl₃(g) and cl₂(g) according to the equation above. a pure sample of pcl₅(g) is placed in a rigid, evacuated 1.00l container. the initial pressure of the pcl₅(g) is 1.00 atm. the temperature is held constant until the pcl₅(g) reaches equilibrium with its decomposition products. the figures below show the initial and equilibrium conditions of the system.
figure 1: initial
pcl₅
p_total = 1.00 atm
figure 2: equilibrium
pcl₅, pcl₃, and cl₂
p_total = 1.40 atm
3 mark for review
as the reaction progresses toward equilibrium, the rate of the forward reaction
a increases until it becomes the same as the reverse reaction rate at equilibrium
b stays constant before and after equilibrium is reached
c decreases to become a constant nonzero rate at equilibrium
d decreases to become zero at equilibrium
For a chemical reaction approaching equilibrium (like \( \text{PCl}_5(g)
ightleftharpoons \text{PCl}_3(g) + \text{Cl}_2(g) \)), the forward reaction rate starts high (as reactant concentration is high) and decreases as reactants are consumed. The reverse reaction rate starts at zero (no products initially) and increases. At equilibrium, the forward and reverse rates become equal (constant, non - zero). So the forward reaction rate decreases to a constant non - zero rate at equilibrium. Option A is wrong because forward and reverse rates become equal, not the same as reverse rate initially. Option B is wrong as the rate changes (forward decreases, reverse increases) until equilibrium. Option D is wrong as the forward rate doesn't become zero at equilibrium (the reaction is dynamic, both forward and reverse occur).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
C. decreases to become a constant nonzero rate at equilibrium