QUESTION IMAGE
Question
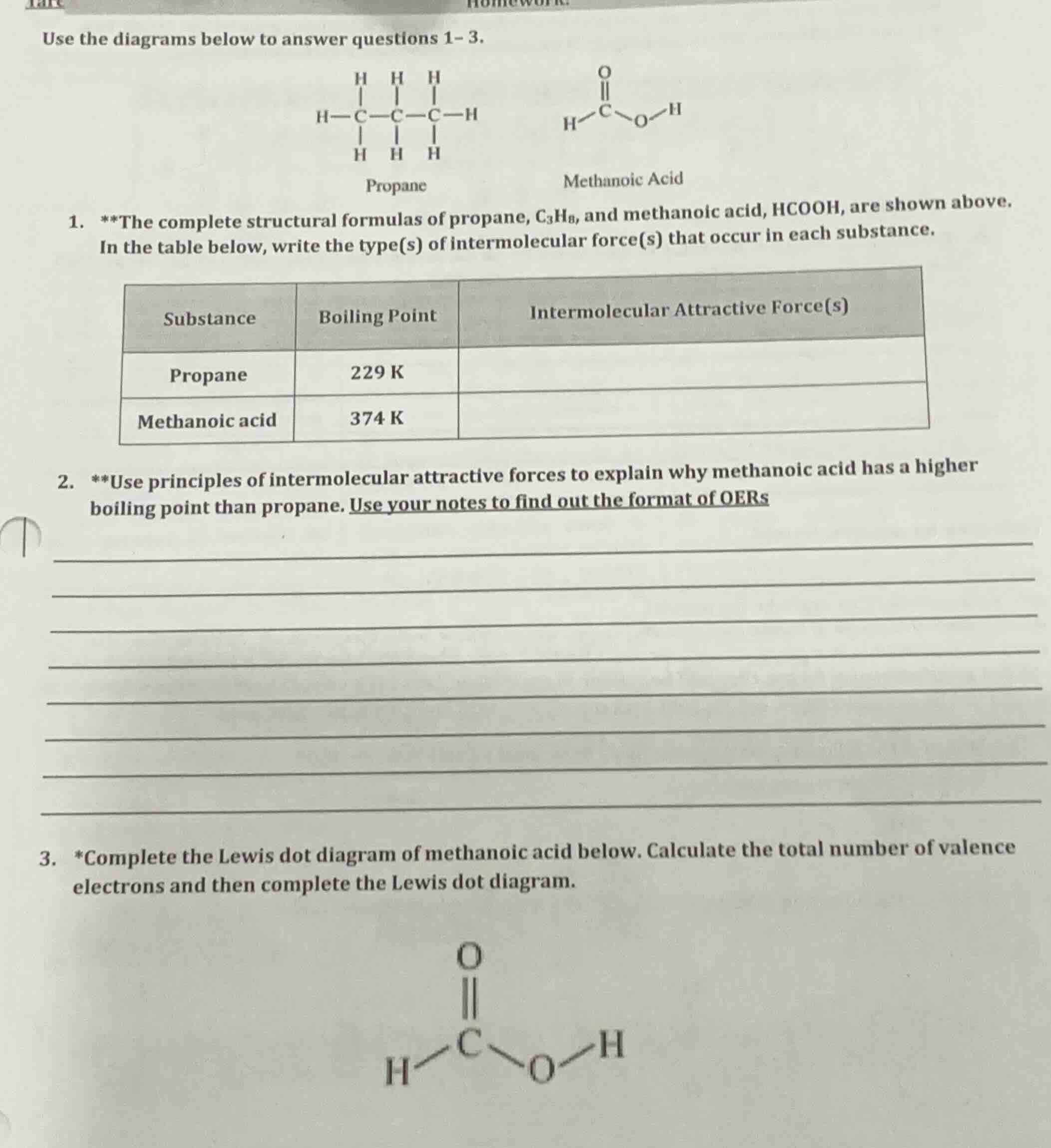
use the diagrams below to answer questions 1- 3.propanemethanoic acid1. the complete structural formulas of propane, $ce{c3h8}$, and methanoic acid, $ce{hcooh}$, are shown above.in the table below, write the type(s) of intermolecular force(s) that occur in each substance.|substance|boiling point|intermolecular attractive force(s)|| ---- | ---- | ---- ||propane|229 k| ||methanoic acid|374 k| |2. use principles of intermolecular attractive forces to explain why methanoic acid has a higher boiling point than propane. use your notes to find out the format of oers3. *complete the lewis dot diagram of methanoic acid below. calculate the total number of valence electrons and then complete the lewis dot diagram.
Step1: Identify forces for propane
Propane ($\text{C}_3\text{H}_8$) is nonpolar, so only London dispersion forces exist between its molecules.
Step2: Identify forces for methanoic acid
Methanoic acid ($\text{HCOOH}$) is polar, has an -OH group, so it has London dispersion forces, dipole-dipole forces, and hydrogen bonding.
Step3: Explain boiling point difference
Stronger intermolecular forces require more energy to overcome. Methanoic acid has strong hydrogen bonding (plus dipole-dipole and London forces), while propane only has weak London dispersion forces. More energy is needed to separate methanoic acid molecules, so it has a higher boiling point.
Step4: Calculate valence electrons for $\text{HCOOH}$
Valence electrons: $\text{H}$ (1) + $\text{C}$ (4) + $2\times\text{O}$ (6 each) + $\text{H}$ (1) = $1+4+12+1=18$ total valence electrons.
Step5: Complete Lewis dot diagram
- The central $\text{C}$ forms a double bond with one $\text{O}$ (uses 4 electrons), a single bond with $\text{H}$ (2 electrons), and a single bond with the other $\text{O}$ (2 electrons).
- The bonded $\text{O}$ forms a single bond with $\text{H}$ (2 electrons).
- Add lone pairs: the double-bonded $\text{O}$ has 2 lone pairs (4 electrons), the single-bonded $\text{O}$ has 2 lone pairs (4 electrons). Total electrons used: $4+2+2+2+4+4=18$, matching the total valence electrons.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
1.
| Substance | Boiling Point | Intermolecular Attractive Force(s) |
|---|---|---|
| Methanoic acid | 374 K | London dispersion forces, dipole-dipole forces, hydrogen bonding |
- Methanoic acid has strong hydrogen bonding (in addition to dipole-dipole and London dispersion forces), while propane only has weak London dispersion forces. Stronger intermolecular forces require more thermal energy to overcome, so methanoic acid has a higher boiling point.
- Total valence electrons in methanoic acid: 18
Lewis dot diagram:
..
H: C::O :O:H
.. ..(Structurally: the double-bonded oxygen has two sets of lone pairs, the single-bonded oxygen (attached to H) has two sets of lone pairs, and all single bonds have their shared electrons accounted for.)