QUESTION IMAGE
Question
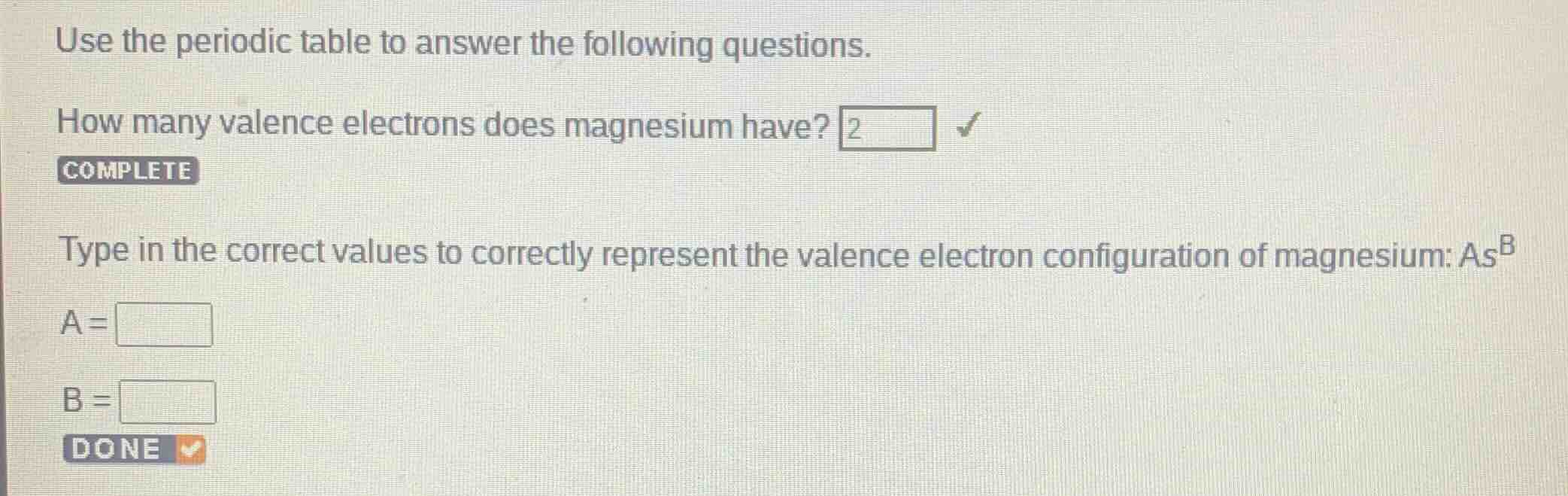
use the periodic table to answer the following questions.
how many valence electrons does magnesium have? 2
complete
type in the correct values to correctly represent the valence electron configuration of magnesium: $a$s$^b$
a =
b =
done
Step1: Find Mg's period/valence shell
Magnesium (Mg) is in period 3, so valence shell is $n=3$.
Step2: Identify valence subshell & electrons
Mg is in group 2, valence electrons occupy the $s$ subshell, with 2 valence electrons.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A = 3
B = 2