QUESTION IMAGE
Question
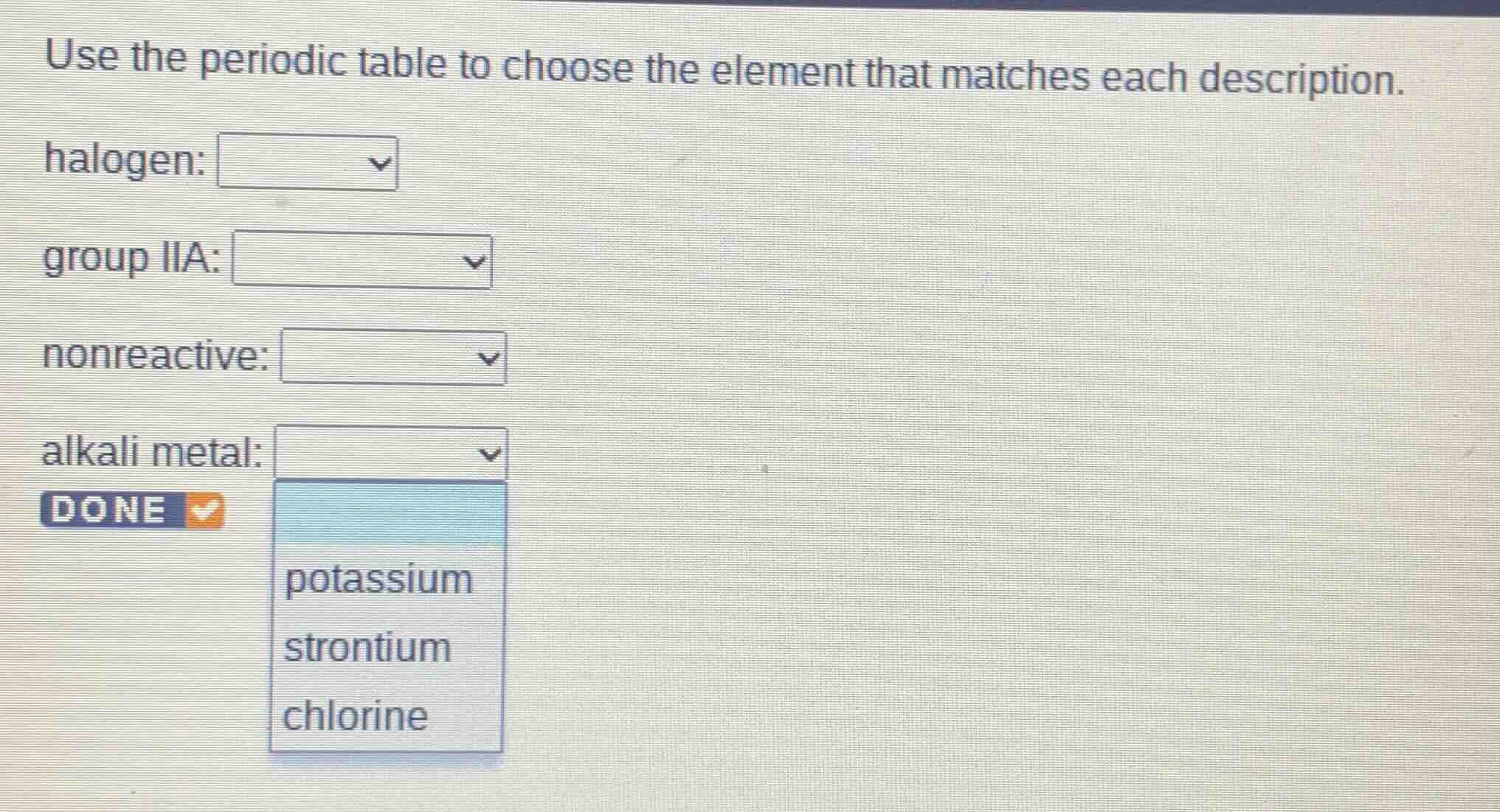
use the periodic table to choose the element that matches each description. halogen: group iia: nonreactive: alkali metal: done potassium strontium chlorine
halogen:
Halogens are in group 17 (VIIA) of the periodic table. Chlorine (Cl) is a halogen. Among the options (potassium, strontium, chlorine), chlorine is the halogen.
Group IIA (alkaline earth metals) includes elements like strontium (Sr). Potassium is group IA, chlorine is group VIIA, so strontium is in group IIA.
Nonreactive elements are noble gases (group 18), but if the options don't include noble gases, maybe a typo or assumption. Wait, the given options for other dropdowns? Wait, the last dropdown (alkali metal) has K, Sr, Cl. Wait, nonreactive should be noble gases, but if no noble gases, maybe the problem expects? Wait, no, maybe the options for nonreactive? Wait, the user's image shows for alkali metal dropdown: potassium, strontium, chlorine. But for nonreactive, noble gases (like helium, neon, argon) are nonreactive. But if the options aren't shown, maybe a mistake. Wait, no, the original problem: maybe the dropdowns have options. Wait, the first dropdown (halogen) should have chlorine (Cl is halogen). Group IIA: strontium (Sr is group IIA). Nonreactive: noble gases (but if options include, say, maybe a mistake, but if we assume the options for nonreactive? Wait, maybe the user's image has more, but in the given, the alkali metal dropdown has K, Sr, Cl. Wait, nonreactive elements are noble gases (group 18), which are inert. If the options for nonreactive are not shown, but maybe a typo. Wait, perhaps the problem's dropdowns: halogen: chlorine (group 17), group IIA: strontium (group 2), nonreactive: maybe a noble gas, but if the options aren't here, but the last dropdown (alkali metal) is potassium (group 1). Wait, let's recheck:
Halogen: group 17, so chlorine (Cl) is halogen.
Group IIA: group 2, strontium (Sr) is group 2.
Nonreactive: noble gases (group 18), but if the options for nonreactive are, say, maybe a mistake, but if we proceed with the given options for other parts. Wait, the alkali metal dropdown: potassium (K) is group 1 (alkali metal), so alkali metal: potassium.
Nonreactive: noble gases (like neon, argon), but if the options aren't shown, maybe the problem expects, but in the given, the user's image for alkali metal dropdown has K, Sr, Cl. So:
nonreactive:
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
chlorine