QUESTION IMAGE
Question
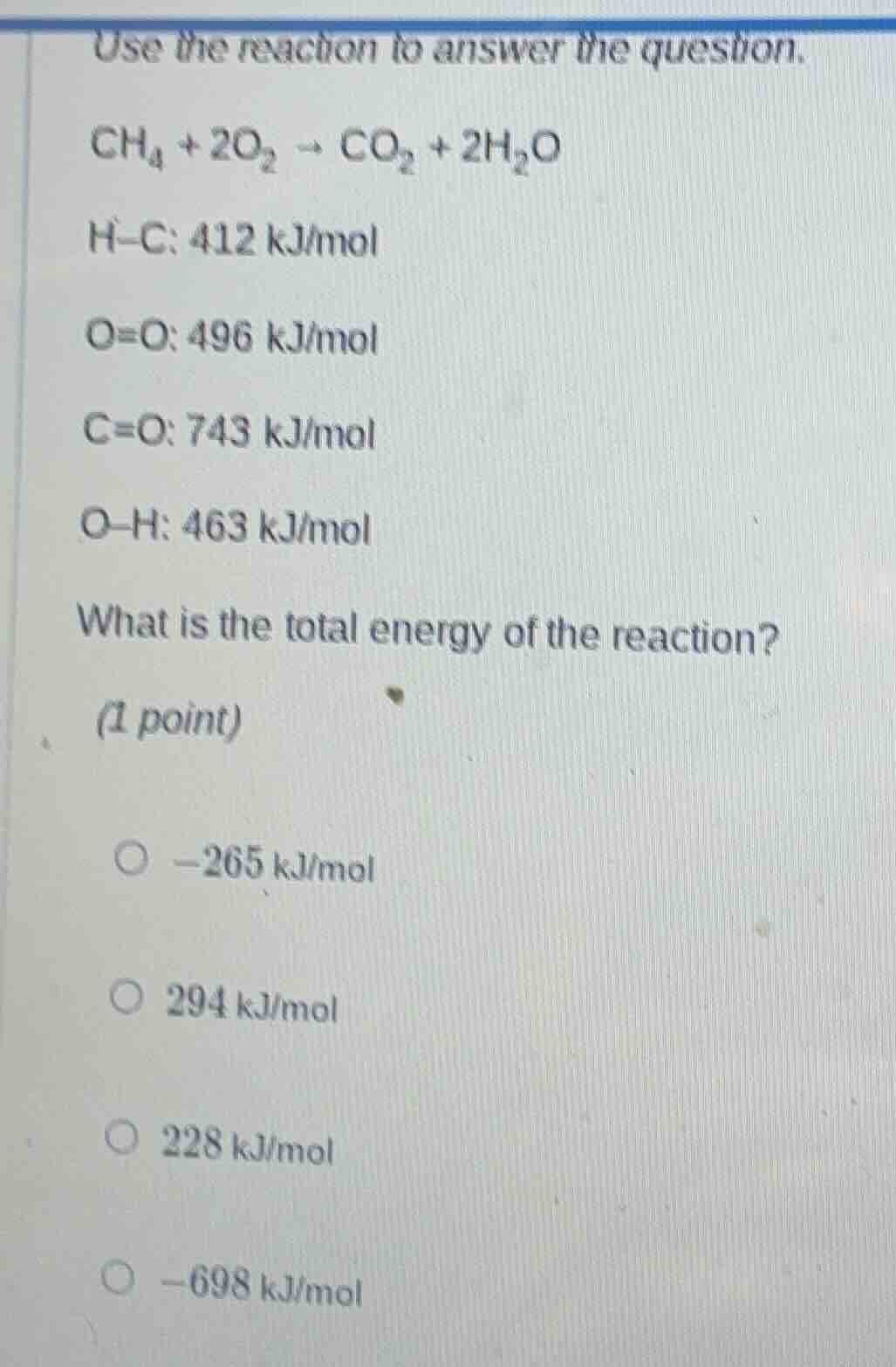
use the reaction to answer the question.
$\ce{ch_{4} + 2o_{2} -> co_{2} + 2h_{2}o}$
h$-$c: 412 kj/mol
o$=$o: 496 kj/mol
c$=$o: 743 kj/mol
o$-$h: 463 kj/mol
what is the total energy of the reaction?
(1 point)
$\boldsymbol{\circ}$ $-265$ kj/mol
$\boldsymbol{\circ}$ $294$ kj/mol
$\boldsymbol{\circ}$ $228$ kj/mol
$\boldsymbol{\circ}$ $-698$ kj/mol
Step1: Calculate energy to break bonds (reactants)
In \( \text{CH}_4 \), there are 4 \( \text{H-C} \) bonds. Energy for \( \text{H-C} \): \( 4 \times 412 = 1648 \) kJ/mol.
In \( 2\text{O}_2 \), there are 2 \( \text{O=O} \) bonds. Energy for \( \text{O=O} \): \( 2 \times 496 = 992 \) kJ/mol.
Total energy to break bonds: \( 1648 + 992 = 2640 \) kJ/mol.
Step2: Calculate energy released from forming bonds (products)
In \( \text{CO}_2 \), there are 2 \( \text{C=O} \) bonds. Energy for \( \text{C=O} \): \( 2 \times 743 = 1486 \) kJ/mol.
In \( 2\text{H}_2\text{O} \), there are 4 \( \text{O-H} \) bonds (2 per \( \text{H}_2\text{O} \)). Energy for \( \text{O-H} \): \( 4 \times 463 = 1852 \) kJ/mol.
Total energy released: \( 1486 + 1852 = 3338 \) kJ/mol.
Step3: Calculate reaction energy (energy released - energy absorbed)
Reaction energy \( \Delta H = \) (energy to break bonds) - (energy to form bonds) \( = 2640 - 3338 = -698 \) kJ/mol? Wait, no—wait, correction: \( \Delta H = \) (energy of bonds broken) - (energy of bonds formed). Wait, actually, \( \Delta H = \sum \text{bonds broken} - \sum \text{bonds formed} \). Wait, let's recheck:
Wait, \( \text{CH}_4 \) (4 H-C), \( 2\text{O}_2 \) (2 O=O) are broken. \( \text{CO}_2 \) (2 C=O), \( 2\text{H}_2\text{O} \) (4 O-H) are formed.
Wait, no: \( \text{CO}_2 \) has two \( \text{C=O} \) bonds (double bonds), so 2 C=O. \( \text{H}_2\text{O} \) has two O-H bonds, so 2 \( \text{H}_2\text{O} \) have 4 O-H.
So bonds broken: \( 4 \times 412 + 2 \times 496 = 1648 + 992 = 2640 \) kJ/mol.
Bonds formed: \( 2 \times 743 + 4 \times 463 = 1486 + 1852 = 3338 \) kJ/mol.
\( \Delta H = 2640 - 3338 = -698 \) kJ/mol. Wait, but let's check again. Wait, maybe I made a mistake in \( \text{CO}_2 \) bonds? Wait, \( \text{CO}_2 \) is \( \text{O=C=O} \), so two double bonds (C=O), so 2 C=O bonds. Correct. \( \text{H}_2\text{O} \) is \( \text{H-O-H} \), two O-H bonds per molecule, so 2 molecules have 4 O-H. Correct.
Wait, but the options include -698 kJ/mol. Wait, but let's recalculate:
Wait, 4412 = 1648; 2496=992; sum broken: 1648+992=2640.
Bonds formed: 2743=1486; 4463=1852; sum formed: 1486+1852=3338.
\( \Delta H = 2640 - 3338 = -698 \) kJ/mol. So that's the answer.
Wait, but let me check again. Maybe I messed up the number of bonds. Let's list all bonds:
Reactants:
- \( \text{CH}_4 \): 4 H-C bonds.
- \( 2\text{O}_2 \): 2 O=O bonds (each \( \text{O}_2 \) has 1 O=O, so 2 molecules have 2 bonds).
Products:
- \( \text{CO}_2 \): 2 C=O bonds (since \( \text{CO}_2 \) is linear, two double bonds).
- \( 2\text{H}_2\text{O} \): each \( \text{H}_2\text{O} \) has 2 O-H bonds, so 2*2=4 O-H bonds.
Yes, that's correct. So energy to break: 4412 + 2496 = 1648 + 992 = 2640.
Energy to form: 2743 + 4463 = 1486 + 1852 = 3338.
\( \Delta H = 2640 - 3338 = -698 \) kJ/mol.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
\(-698\) kJ/mol (corresponding to the option: \(-698\) kJ/mol)