QUESTION IMAGE
Question
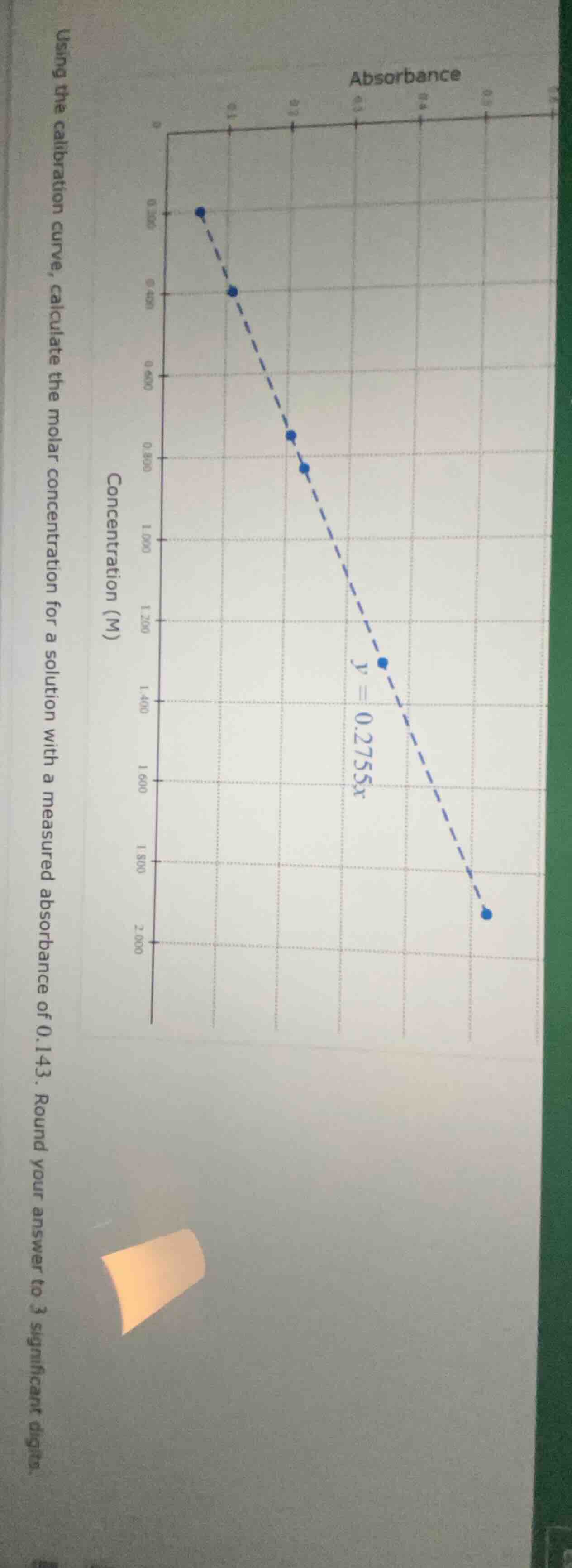
using the calibration curve, calculate the molar concentration for a solution with a measured absorbance of 0.143. round your answer to 3 significant digits.
(chart: absorbance on x - axis, concentration (m) on y - axis, calibration curve equation $y = 0.2755x$)
Step1: Identify the formula
The calibration curve is given by \( y = 0.2755x \), where \( y \) is the absorbance and \( x \) is the molar concentration (M). We know the absorbance \( y = 0.143 \), and we need to find \( x \).
Step2: Rearrange the formula to solve for \( x \)
From \( y = 0.2755x \), we can solve for \( x \) by dividing both sides by \( 0.2755 \):
\[ x=\frac{y}{0.2755} \]
Step3: Substitute the value of \( y \)
Substitute \( y = 0.143 \) into the formula:
\[ x=\frac{0.143}{0.2755} \]
Step4: Calculate the value
Perform the division:
\[ x\approx0.519 \] (rounded to 3 significant digits)
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The molar concentration is approximately \(\boldsymbol{0.519}\) M.