QUESTION IMAGE
Question
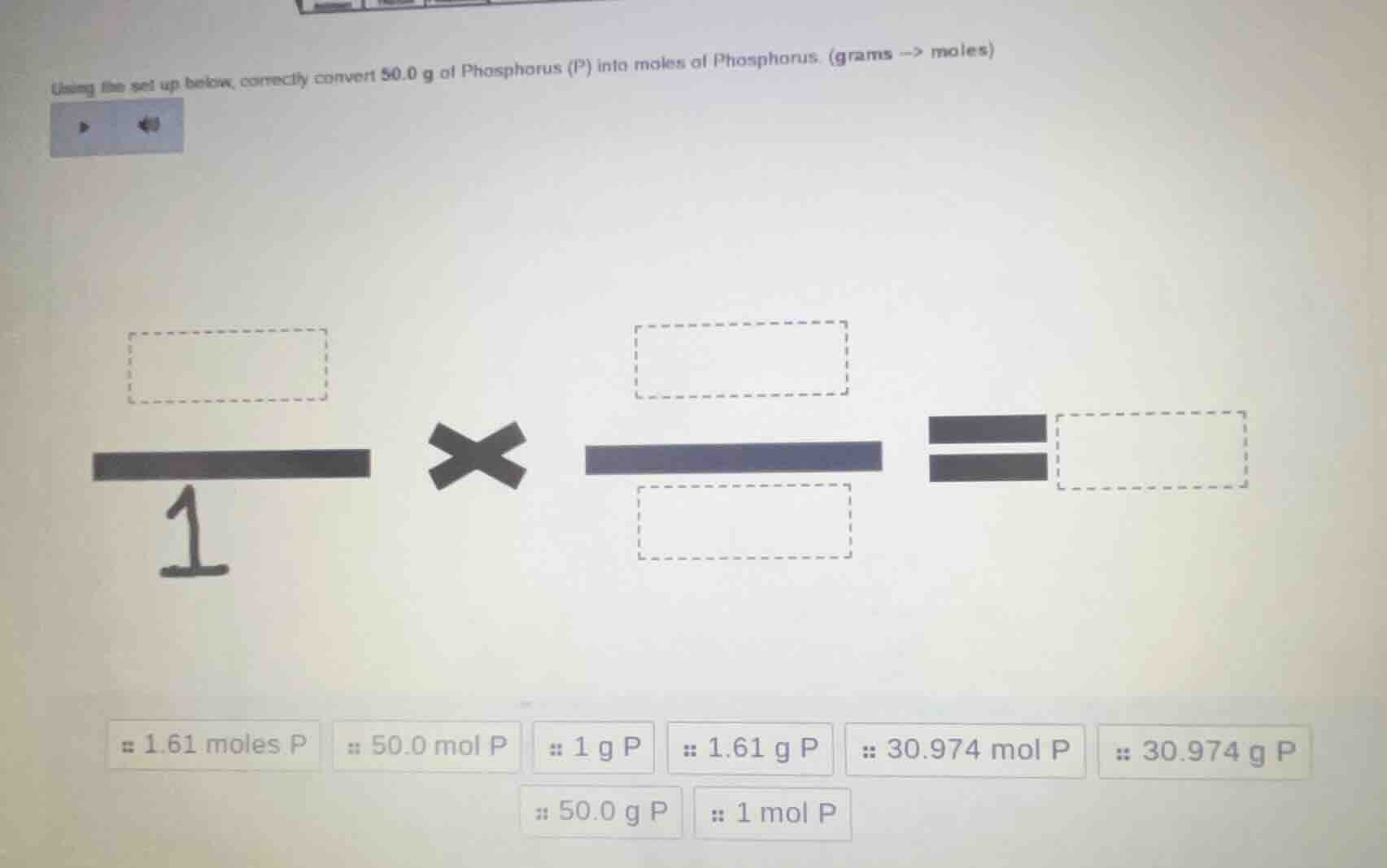
using the set up below, correctly convert 50.0 g of phosphorus (p) into moles of phosphorus. (grams -> moles) 1 × ( )/( ) = ( ) 1.61 moles p 50.0 mol p 1 g p 1.61 g p 30.974 mol p 30.974 g p 50.0 g p 1 mol p
Step1: Identify given mass and molar mass
We have 50.0 g of P. The molar mass of P is 30.974 g/mol (from the options, 30.974 g P corresponds to 1 mol P). So the first box (numerator of first fraction) is 50.0 g P, the second fraction's numerator is 1 mol P, and denominator is 30.974 g P.
$$\frac{50.0\ \text{g P}}{1} \times \frac{1\ \text{mol P}}{30.974\ \text{g P}}$$
Step2: Perform the calculation
Calculate \( \frac{50.0}{30.974} \approx 1.61 \) moles of P.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
First box: 50.0 g P
Second fraction numerator: 1 mol P
Second fraction denominator: 30.974 g P
Result: 1.61 moles P
So the setup is \(\frac{50.0\ \text{g P}}{1} \times \frac{1\ \text{mol P}}{30.974\ \text{g P}} = 1.61\ \text{moles P}\)