QUESTION IMAGE
Question
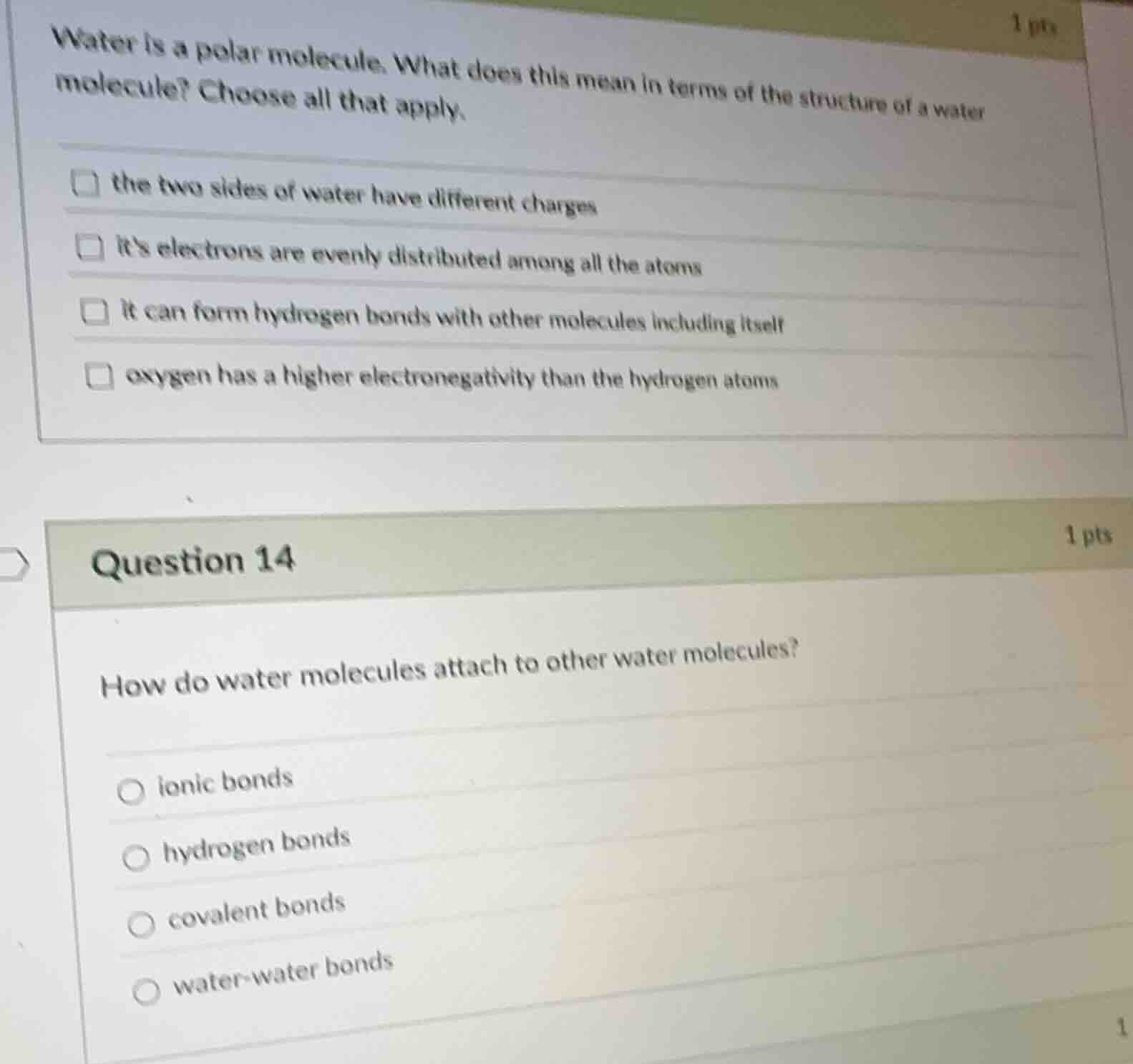
water is a polar molecule. what does this mean in terms of the structure of a water molecule? choose all that apply. the two sides of water have different charges its electrons are evenly distributed among all the atoms it can form hydrogen bonds with other molecules including itself oxygen has a higher electronegativity than the hydrogen atoms question 14 1 pts how do water molecules attach to other water molecules? ionic bonds hydrogen bonds covalent bonds water-water bonds
Response
First Question (Water as a polar molecule):
Brief Explanations
- For "the two sides of water have different charges": A polar molecule has a partial positive and partial negative side due to unequal electron sharing, so this applies.
- For "It's electrons are evenly distributed among all the atoms": In polar molecules, electrons are unevenly distributed (due to electronegativity differences), so this is incorrect.
- For "It can form hydrogen bonds with other molecules including itself": Polar molecules (like water with partial charges) can form hydrogen bonds, so this applies.
- For "oxygen has a higher electronegativity than the hydrogen atoms": Oxygen's higher electronegativity causes unequal electron sharing, making water polar, so this applies.
Brief Explanations
- Ionic bonds: Form between ions (charged atoms), not between water molecules.
- Hydrogen bonds: Water molecules have partial charges (polar), so the partial positive H of one water bonds with partial negative O of another via hydrogen bonds.
- Covalent bonds: Involve sharing electrons between atoms (e.g., within a water molecule, H - O bonds), not between separate water molecules.
- Water - water bonds: Not a recognized bond type.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- the two sides of water have different charges
- it can form hydrogen bonds with other molecules including itself
- oxygen has a higher electronegativity than the hydrogen atoms