QUESTION IMAGE
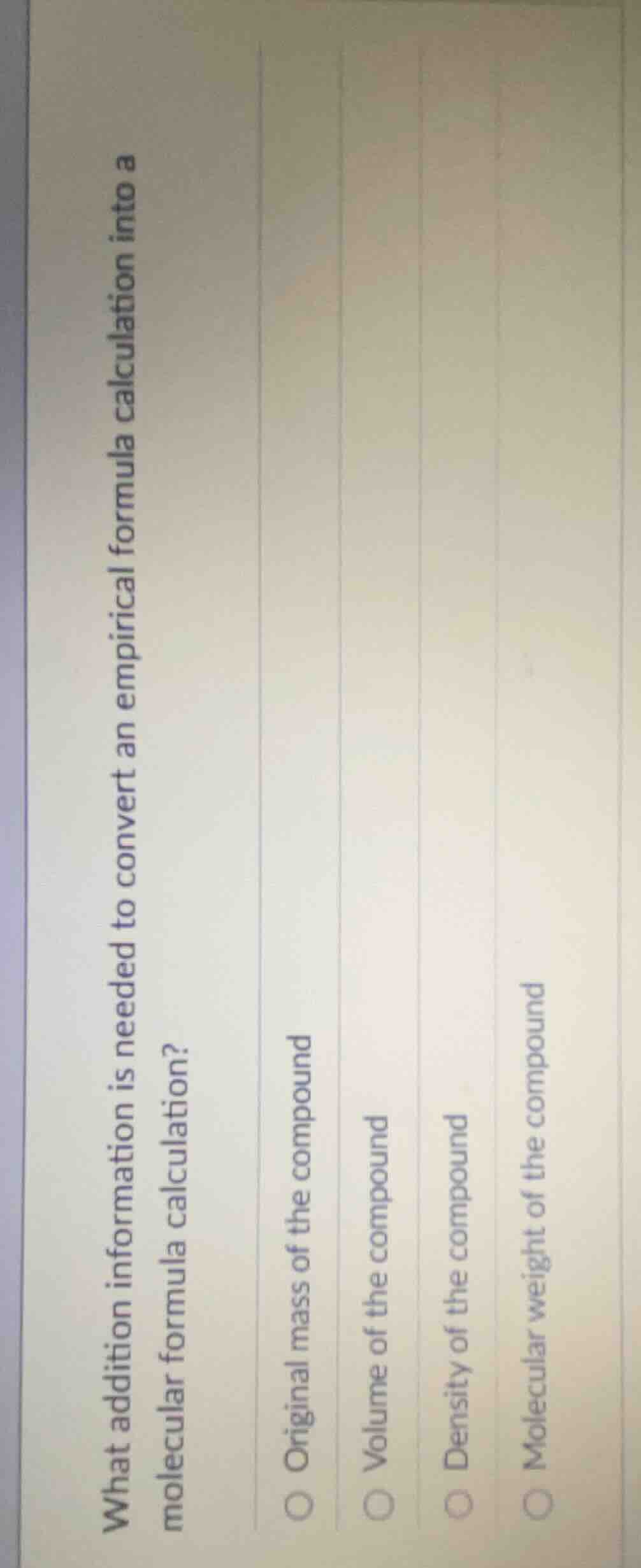
Question
what addition information is needed to convert an empirical formula calculation into a molecular formula calculation?
original mass of the compound
volume of the compound
density of the compound
molecular weight of the compound
The empirical formula gives the simplest whole-number ratio of atoms in a compound, while the molecular formula gives the actual number of each type of atom. To convert the empirical formula to the molecular formula, you need to find the ratio of the compound's molecular weight to the empirical formula weight, then multiply each subscript in the empirical formula by this ratio. Other values like original mass, volume, or density do not provide the necessary information to scale the empirical formula to the molecular formula.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. Molecular weight of the compound