QUESTION IMAGE
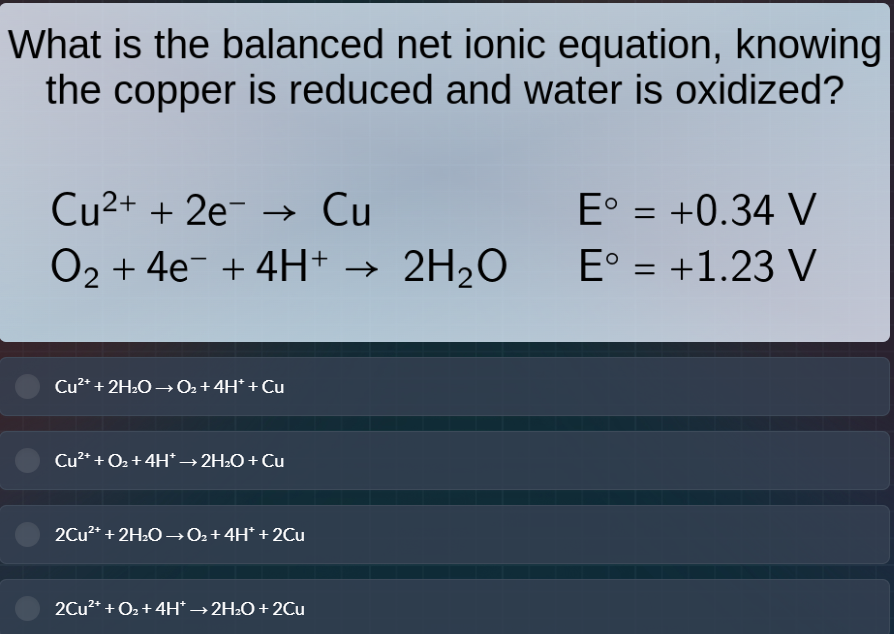
Question
what is the balanced net ionic equation, knowing the copper is reduced and water is oxidized?
cu²⁺ + 2e⁻ → cu e° = +0.34 v
o₂ + 4e⁻ + 4h⁺ → 2h₂o e° = +1.23 v
options:
cu²⁺ + 2h₂o → o₂ + 4h⁺ + cu
cu²⁺ + o₂ + 4h⁺ → 2h₂o + cu
2cu²⁺ + 2h₂o → o₂ + 4h⁺ + 2cu
2cu²⁺ + o₂ + 4h⁺ → 2h₂o + 2cu
Step1: Identify Oxidation and Reduction Half - Reactions
- Reduction: The process where a species gains electrons. Given that copper is reduced, the reduction half - reaction is $\ce{Cu^{2+} + 2e^- \to Cu}$ with $E^{\circ}= + 0.34\ V$.
- Oxidation: The process where a species loses electrons. Given that water is oxidized, the oxidation half - reaction is the reverse of $\ce{O_{2}+4e^- + 4H^{+}\to 2H_{2}O}$. So the oxidation half - reaction is $\ce{2H_{2}O\to O_{2}+4H^{+}+4e^-}$. The standard reduction potential of the reverse reaction (oxidation) will have the opposite sign, but for balancing electrons, we focus on the electron transfer.
Step2: Balance the Number of Electrons Transferred
- The reduction half - reaction ( $\ce{Cu^{2+} + 2e^- \to Cu}$) involves the transfer of 2 electrons per mole of $\ce{Cu^{2+}}$.
- The oxidation half - reaction ( $\ce{2H_{2}O\to O_{2}+4H^{+}+4e^-}$) involves the transfer of 4 electrons per mole of $\ce{O_{2}}$ produced.
- To balance the number of electrons, we multiply the reduction half - reaction by 2. So the modified reduction half - reaction is $2\times(\ce{Cu^{2+} + 2e^- \to Cu})$, which gives $\ce{2Cu^{2+}+4e^-\to 2Cu}$.
Step3: Add the Oxidation and Modified Reduction Half - Reactions
- Oxidation half - reaction: $\ce{2H_{2}O\to O_{2}+4H^{+}+4e^-}$
- Modified reduction half - reaction: $\ce{2Cu^{2+}+4e^-\to 2Cu}$
- When we add these two half - reactions together, the electrons ($4e^-$) cancel out. We get:
$\ce{2Cu^{2+}+2H_{2}O\to O_{2}+4H^{+}+2Cu}$
Step4: Check the Options
- Let's analyze each option:
- Option 1: $\ce{Cu^{2+}+2H_{2}O\to O_{2}+4H^{+}+Cu}$: The number of electrons is not balanced. The reduction of $\ce{Cu^{2+}}$ (2 electrons per $\ce{Cu^{2+}}$) and oxidation of $\ce{H_{2}O}$ (4 electrons per $\ce{O_{2}}$) are not balanced here.
- Option 2: $\ce{Cu^{2+}+O_{2}+4H^{+}\to 2H_{2}O + Cu}$: This reaction shows $\ce{O_{2}}$ being reduced (since it is on the reactant side and is gaining electrons) and $\ce{Cu^{2+}}$ being reduced, which is incorrect as water should be oxidized.
- Option 3: $\ce{2Cu^{2+}+2H_{2}O\to O_{2}+4H^{+}+2Cu}$: This reaction has the correct number of electrons balanced. The oxidation of $\ce{H_{2}O}$ (to $\ce{O_{2}}$) and reduction of $\ce{Cu^{2+}}$ (to $\ce{Cu}$) are balanced in terms of electron transfer (4 electrons from oxidation of 2 moles of $\ce{H_{2}O}$ and 4 electrons from reduction of 2 moles of $\ce{Cu^{2+}}$).
- Option 4: $\ce{2Cu^{2+}+O_{2}+4H^{+}\to 2H_{2}O + 2Cu}$: This reaction shows $\ce{O_{2}}$ being reduced (on the reactant side) and $\ce{Cu^{2+}}$ being reduced, which is incorrect as water should be oxidized.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$\boldsymbol{2\ce{Cu^{2+}} + 2\ce{H_{2}O}\to \ce{O_{2}} + 4\ce{H^{+}} + 2\ce{Cu}}$ (corresponding to the option "2Cu²⁺ + 2H₂O → O₂ + 4H⁺ + 2Cu")