QUESTION IMAGE
Question
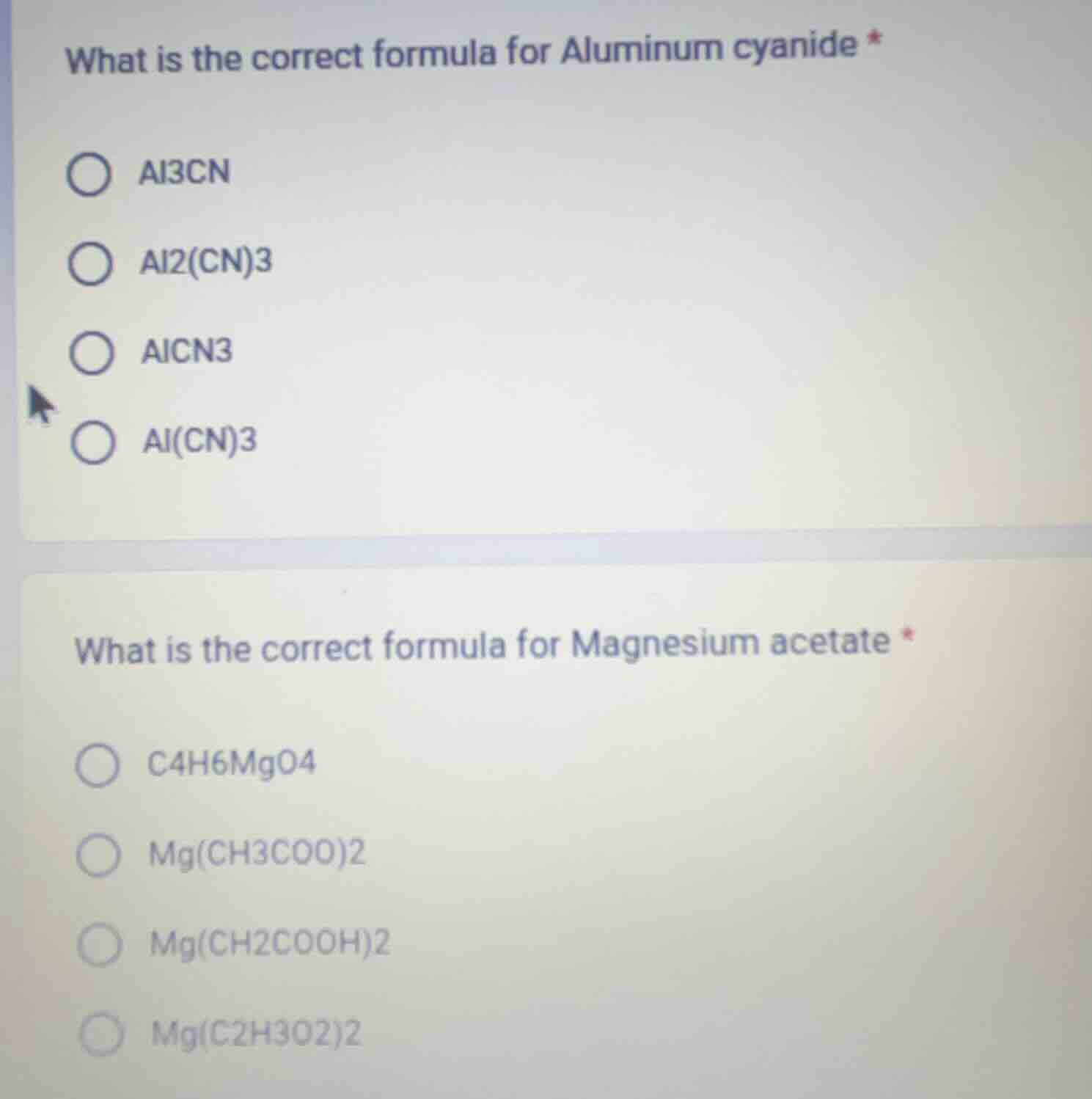
what is the correct formula for aluminum cyanide *
al3cn
al2(cn)3
alcn3
al(cn)3
what is the correct formula for magnesium acetate *
c4h6mgo4
mg(ch3coo)2
mg(ch2cooh)2
mg(c2h3o2)2
First Question: Correct formula for Aluminum cyanide
Step1: Determine ion charges
Aluminum (Al) has a charge of \( +3 \) (group 13 metal, loses 3 electrons). Cyanide (\( \text{CN}^- \)) has a charge of \( -1 \) (polyatomic ion, charge -1).
Step2: Balance charges
To balance the charges, the number of \( \text{CN}^- \) ions needed to neutralize \( \text{Al}^{3+} \) is calculated by the formula: \( \text{Charge of Al} \times \text{number of Al} = \text{Charge of CN}^- \times \text{number of CN}^- \). Let \( x \) be the number of Al and \( y \) be the number of \( \text{CN}^- \). So \( 3x = 1y \). For a neutral compound, the simplest ratio is \( x = 1 \), \( y = 3 \). Thus, the formula is \( \text{Al}(\text{CN})_3 \).
Step1: Determine ion charges
Magnesium (Mg) has a charge of \( +2 \) (group 2 metal, loses 2 electrons). Acetate (\( \text{CH}_3\text{COO}^- \)) has a charge of \( -1 \) (polyatomic ion, charge -1).
Step2: Balance charges
To balance the charges, let \( x \) be the number of Mg and \( y \) be the number of acetate ions. So \( 2x = 1y \). The simplest ratio is \( x = 1 \), \( y = 2 \). The formula for acetate is \( \text{CH}_3\text{COO}^- \), so the formula for magnesium acetate is \( \text{Mg}(\text{CH}_3\text{COO})_2 \) (or \( \text{Mg(C}_2\text{H}_3\text{O}_2\text{)}_2 \) since \( \text{CH}_3\text{COO}^- \) can be written as \( \text{C}_2\text{H}_3\text{O}_2^- \)).
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. \( \text{Al(CN)}_3 \) (assuming the options are labeled as A: Al3CN, B: Al2(CN)3, C: AlCN3, D: Al(CN)3)