QUESTION IMAGE
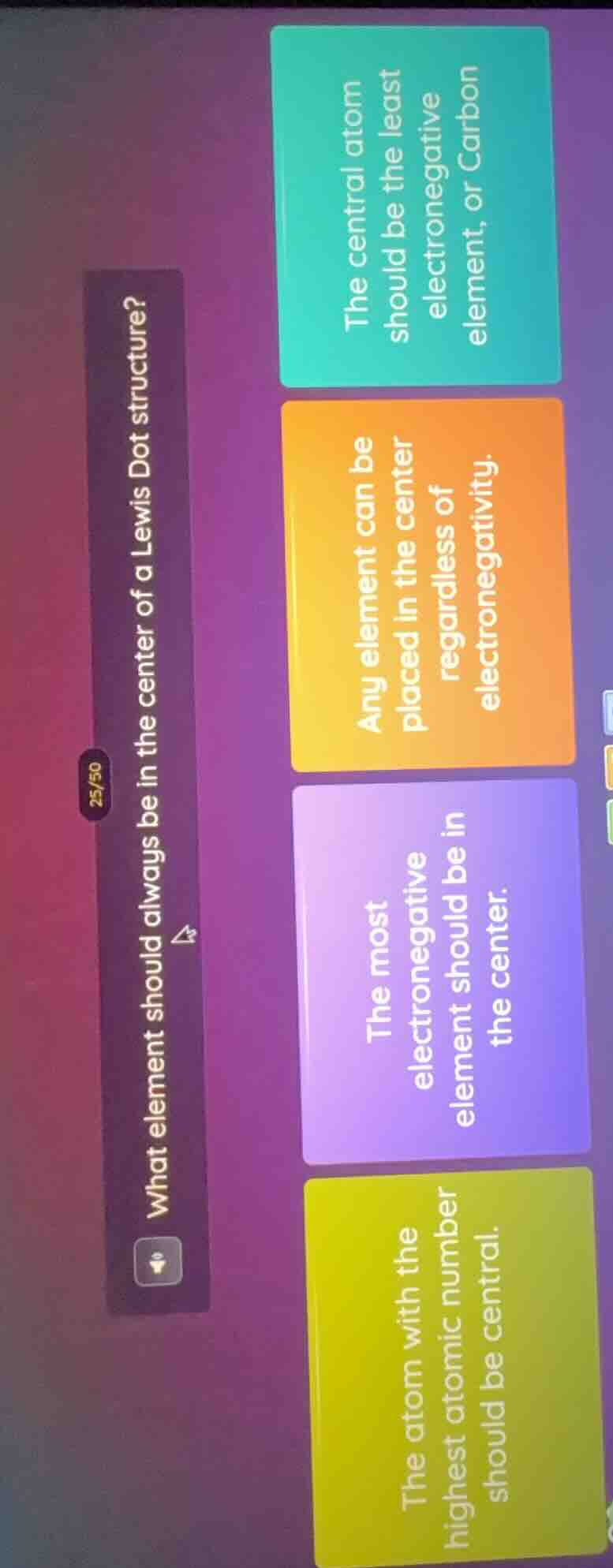
Question
what element should always be in the center of a lewis dot structure?
the central atom should be the least electronegative element, or carbon
any element can be placed in the center regardless of electronegativity.
the most electronegative element should be in the center.
the atom with the highest atomic number should be central.
Lewis Dot structure rules dictate that the central atom is typically the least electronegative element (with the exception of hydrogen, which is never central), as it can form more bonds to surround itself with other atoms. Carbon is a very common central atom because it is moderately electronegative and forms four stable bonds, fitting this guideline. The other options contradict standard Lewis structure conventions.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
The central atom should be the least electronegative element, or Carbon