QUESTION IMAGE
Question
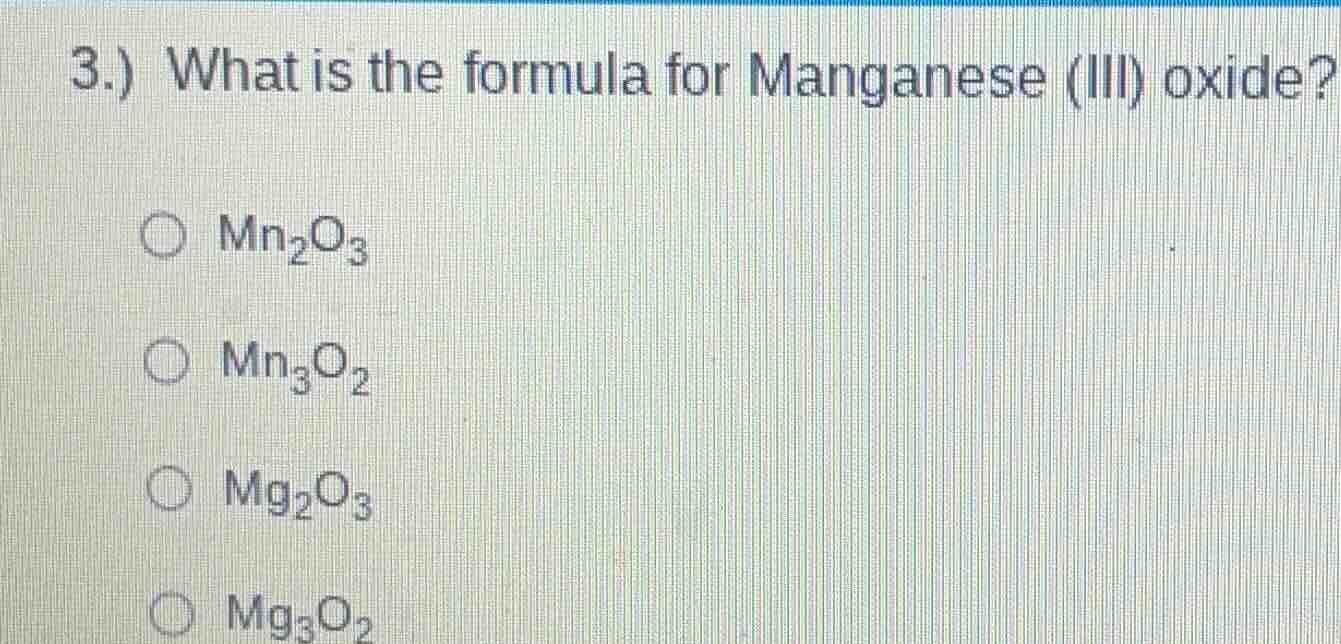
3.) what is the formula for manganese (iii) oxide?
○ mn₂o₃
○ mn₃o₂
○ mg₂o₃
○ mg₃o₂
Step1: Identify the ions
Manganese (III) means the manganese ion has a charge of \(+3\) (\(Mn^{3+}\)), and oxide is the oxide ion with a charge of \(-2\) (\(O^{2-}\)).
Step2: Use the criss - cross method
To form a neutral compound, the number of positive charges must equal the number of negative charges. For \(Mn^{3+}\) and \(O^{2-}\), we cross - multiply the charges. The charge of Mn (\(3\)) becomes the subscript of O, and the charge of O (\(2\)) becomes the subscript of Mn. So the formula should be \(Mn_2O_3\) (since \(3\times2 = 2\times3=6\), the total positive charge \(2\times( + 3)=+6\) and total negative charge \(3\times(- 2)=-6\), making the compound neutral). Also, we can eliminate options with Mg (magnesium) as the question is about manganese oxide.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
A. \(Mn_2O_3\)