QUESTION IMAGE
Question
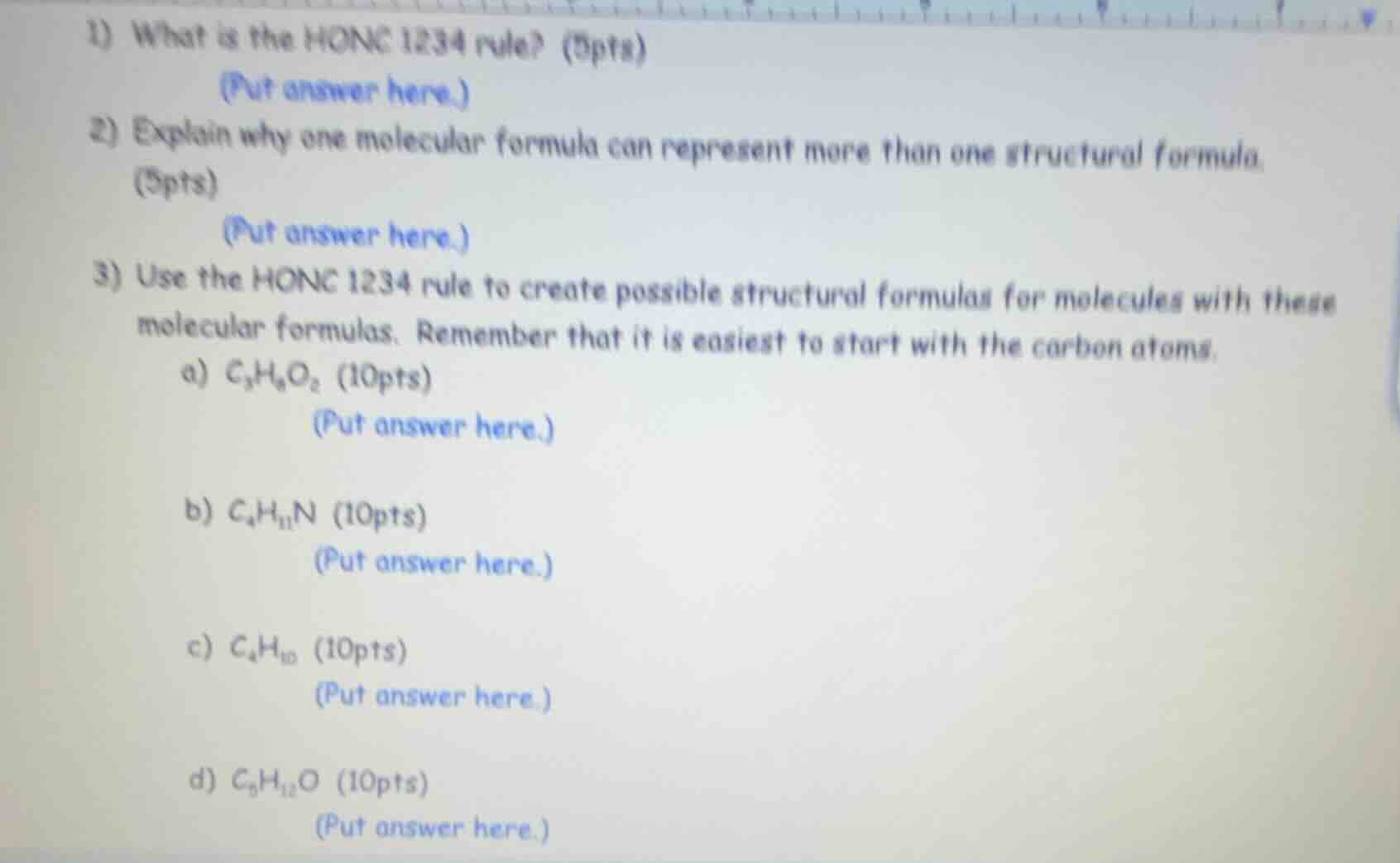
- what is the honc 1234 rule? (5pts)(put answer here.)2) explain why one molecular formula can represent more than one structural formula. (5pts)(put answer here.)3) use the honc 1234 rule to create possible structural formulas for molecules with these molecular formulas. remember that it is easiest to start with the carbon atoms.a) $ce{c3h6o2}$ (10pts)(put answer here.)b) $ce{c4h11n}$ (10pts)(put answer here.)c) $ce{c4h10}$ (10pts)(put answer here.)d) $ce{c5h12o}$ (10pts)(put answer here.)
Brief Explanations
- The HONC 1234 rule is a guideline for covalent bonding in organic molecules, stating the typical number of covalent bonds each element forms to reach a full valence shell.
- One molecular formula can correspond to multiple structural formulas due to isomerism, where atoms can be arranged in different bonding patterns while maintaining the same number and type of atoms.
- For each molecular formula, the HONC rule (H=1 bond, O=2 bonds, N=3 bonds, C=4 bonds) is used to draw valid structural arrangements, prioritizing carbon skeletons first, then adding other atoms to satisfy bonding rules.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
- The HONC 1234 rule is a bonding guideline for common organic elements: Hydrogen (H) forms 1 covalent bond, Oxygen (O) forms 2 covalent bonds, Nitrogen (N) forms 3 covalent bonds, and Carbon (C) forms 4 covalent bonds to achieve a stable valence electron configuration.
- This occurs due to structural isomerism: molecules with the same molecular formula can have different arrangements of atoms (different bonding connections between atoms), creating distinct structural formulas with different properties.
3)
a) $C_3H_6O_2$:
- Propanoic acid: $CH_3CH_2COOH$ (structure: $H_3C-CH_2-\overset{O}{\overset{||}{C}}-OH$)
- Methyl acetate: $CH_3COOCH_3$ (structure: $H_3C-\overset{O}{\overset{||}{C}}-O-CH_3$)
b) $C_4H_{11}N$:
- Butan-1-amine: $CH_3CH_2CH_2CH_2NH_2$
- Butan-2-amine: $CH_3CH_2CH(NH_2)CH_3$
- N-Methylpropan-1-amine: $CH_3CH_2CH_2NHCH_3$
c) $C_4H_{10}$:
- Butane: $CH_3CH_2CH_2CH_3$
- 2-Methylpropane: $(CH_3)_3CH$
d) $C_5H_{12}O$:
- Pentan-1-ol: $CH_3CH_2CH_2CH_2CH_2OH$
- 2-Methylbutan-1-ol: $(CH_3)_2CHCH_2CH_2OH$
- 3-Methylbutan-1-ol: $CH_3CH_2CH(CH_3)CH_2OH$