QUESTION IMAGE
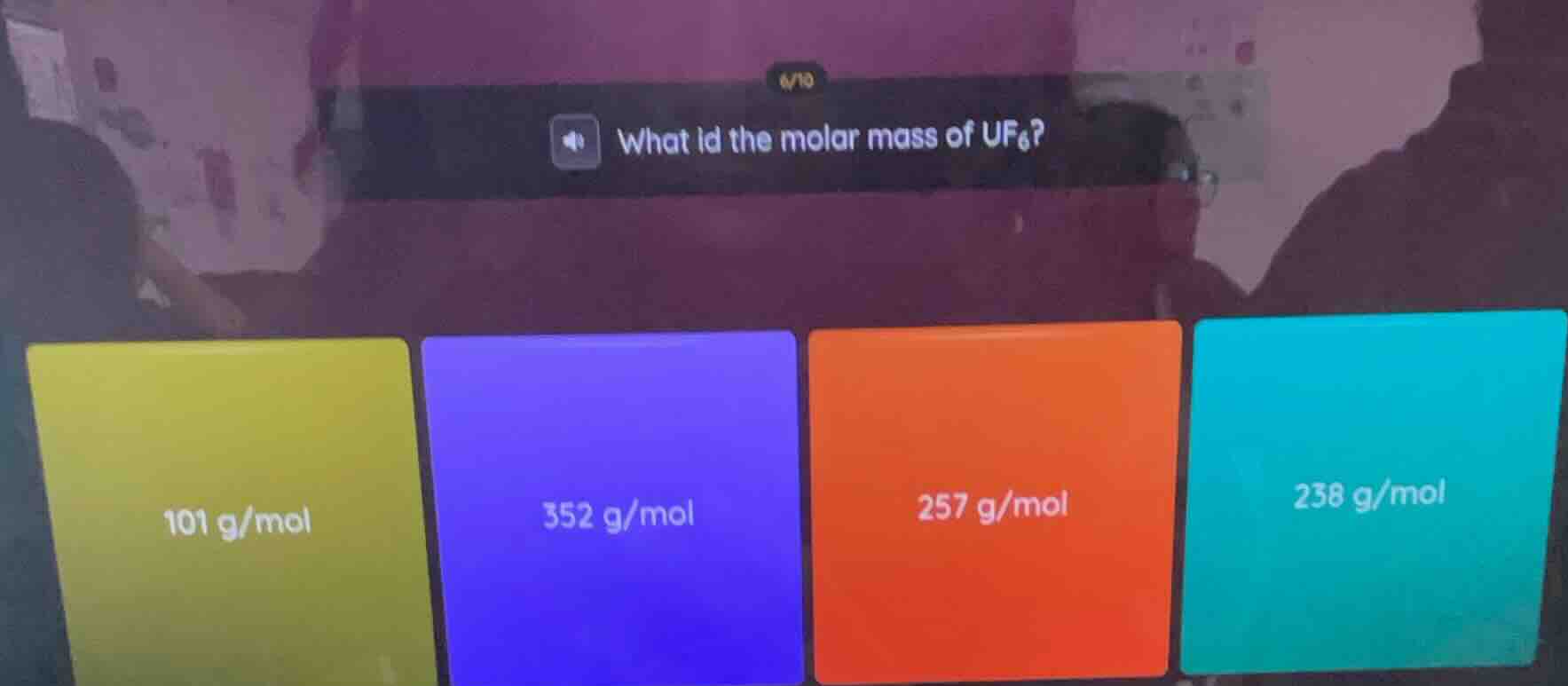
Question
what id the molar mass of uf₆?
101 g/mol
352 g/mol
257 g/mol
238 g/mol
Step1: Find molar mass of U
Molar mass of U (Uranium) is $238.03\ \text{g/mol}$.
Step2: Find molar mass of F
Molar mass of F (Fluorine) is $19.00\ \text{g/mol}$.
Step3: Calculate molar mass of $\text{UF}_6$
In $\text{UF}_6$, there is 1 U and 6 F. So molar mass = $238.03 + 6\times19.00$.
Calculate $6\times19.00 = 114.00$.
Then $238.03 + 114.00 = 352.03\ \text{g/mol}$, approximately $352\ \text{g/mol}$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
352 g/mol (corresponding to the purple option: 352 g/mol)