QUESTION IMAGE
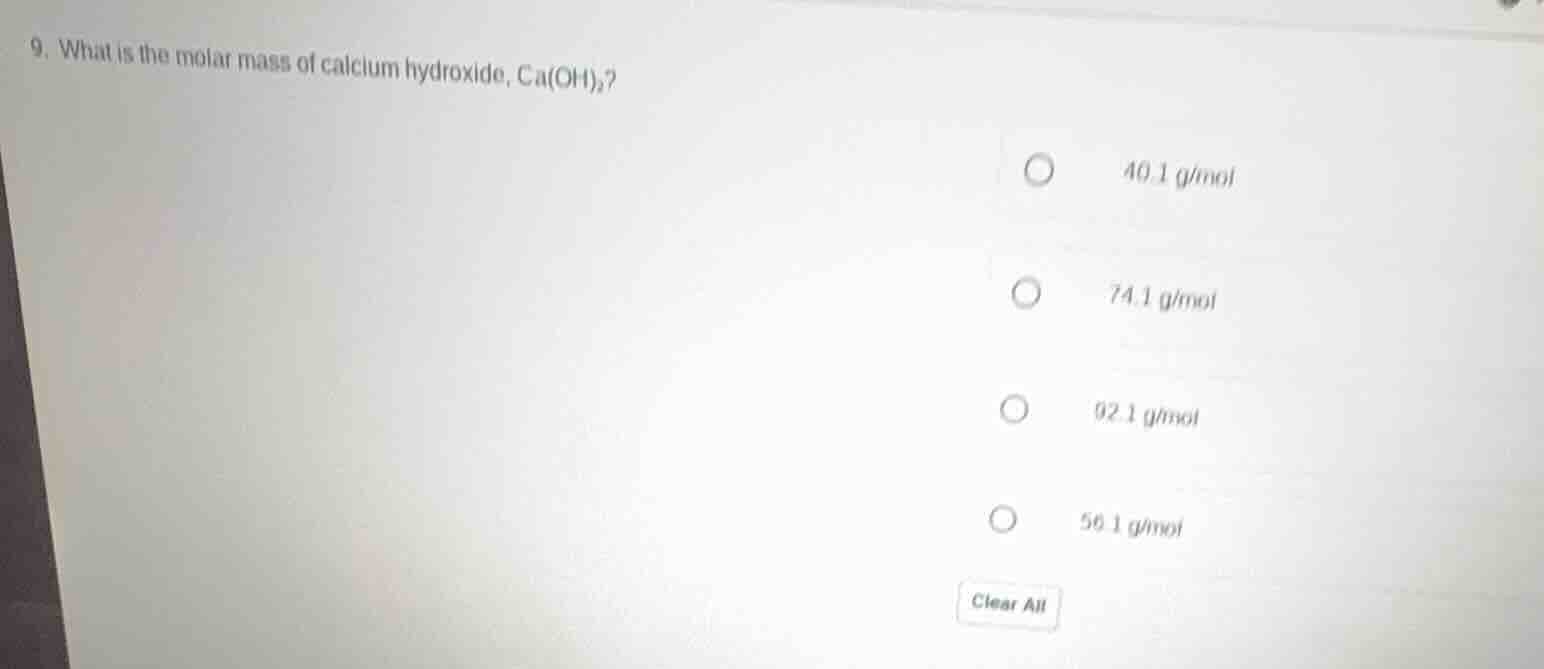
Question
- what is the molar mass of calcium hydroxide, ca(oh)₂?
40.1 g/mol
74.1 g/mol
92.1 g/mol
56.1 g/mol
clear all
Step1: Identify atomic masses
Atomic mass of Ca: $40.1\ g/mol$, O: $16.0\ g/mol$, H: $1.0\ g/mol$.
Step2: Calculate for each atom
In $\ce{Ca(OH)2}$, there is 1 Ca, 2 O, 2 H.
Mass from Ca: $1\times40.1 = 40.1\ g/mol$.
Mass from O: $2\times16.0 = 32.0\ g/mol$.
Mass from H: $2\times1.0 = 2.0\ g/mol$.
Step3: Sum the masses
Total molar mass: $40.1 + 32.0 + 2.0 = 74.1\ g/mol$.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
74.1 g/mol (corresponding to the option with "74.1 g/mol")