QUESTION IMAGE
Question
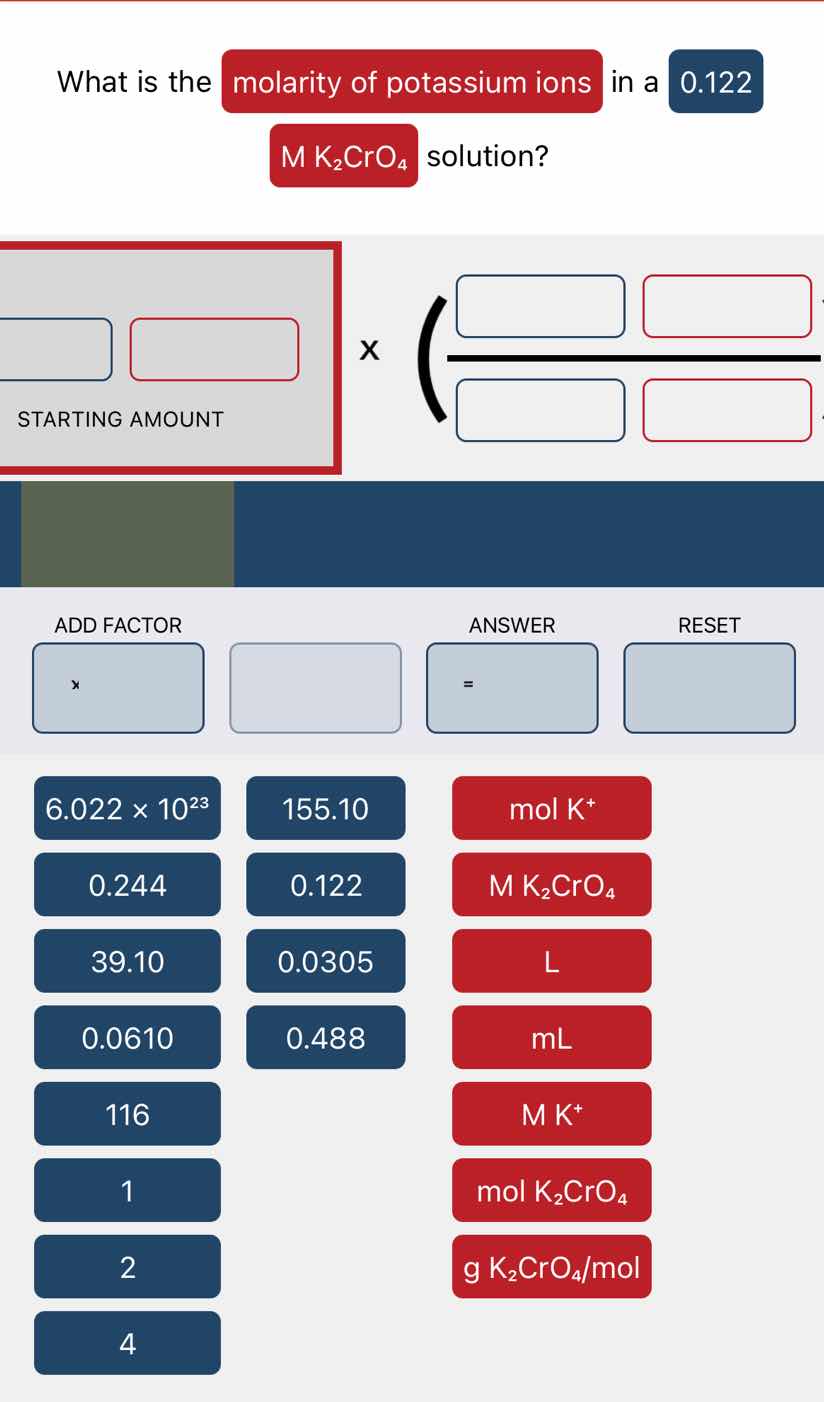
what is the molarity of potassium ions in a 0.122 m k₂cro₄ solution?
Step1: Identify starting molarity
Starting amount: $0.122\ \text{M } K_2CrO_4$
Step2: Use ion mole ratio
From $K_2CrO_4$, ratio: $\frac{2\ \text{mol } K^+}{1\ \text{mol } K_2CrO_4}$
Step3: Calculate $K^+$ molarity
$0.122\ \text{M } K_2CrO_4 \times \frac{2\ \text{mol } K^+}{1\ \text{mol } K_2CrO_4}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
$0.244\ \text{M } K^+$