QUESTION IMAGE
Question
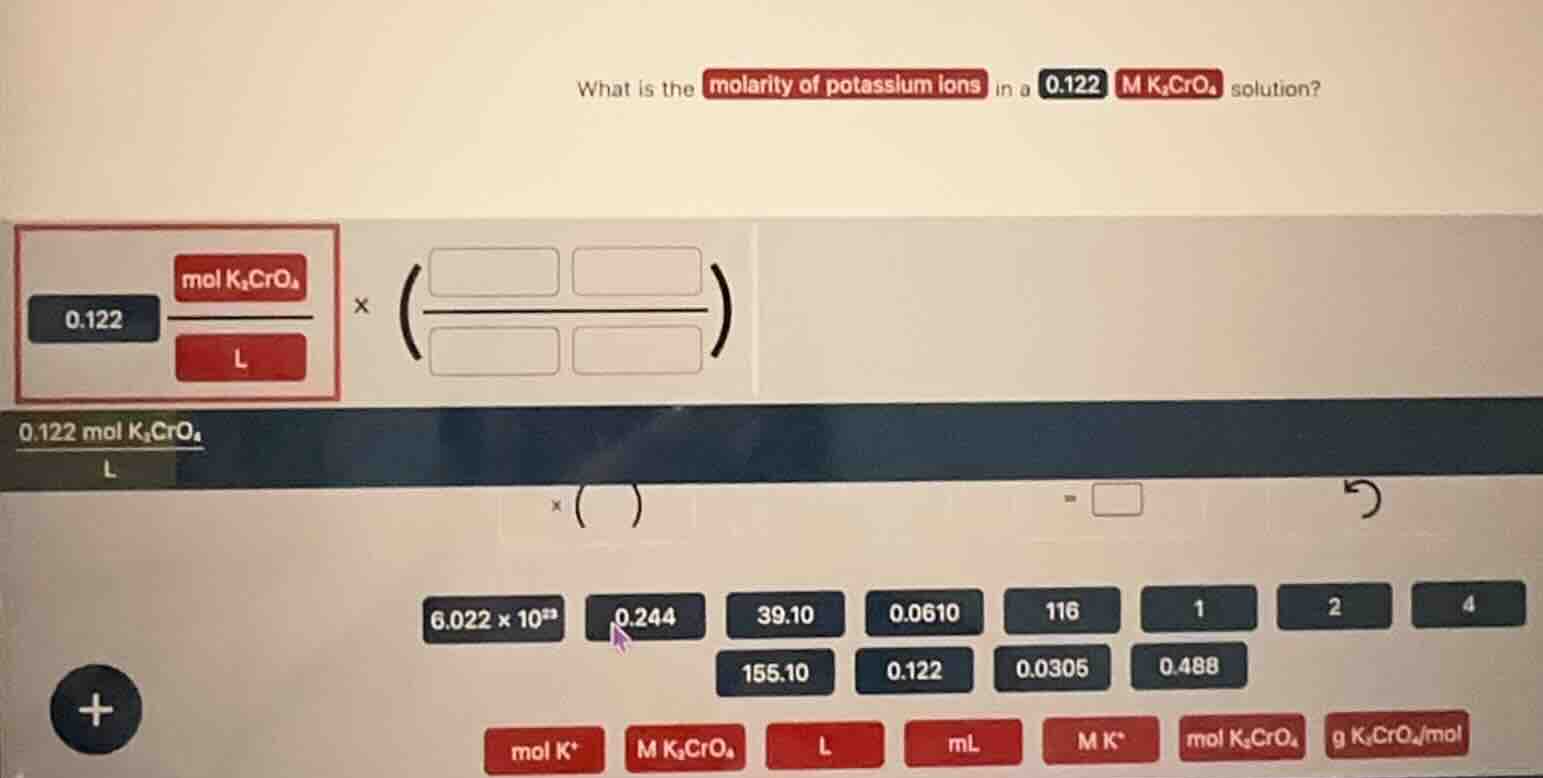
what is the molarity of potassium ions in a 0.122 m k₂cro₄ solution? 0.122 (mol k₂cro₄ / l) × ( / ) 0.122 mol k₂cro₄ / l × ( ) = options: 6.022×10²³, 0.244, 39.10, 0.0610, 116, 1, 2, 4, 155.10, 0.122, 0.0305, 0.488; labels: mol k⁺, m k₂cro₄, l, ml, m k⁺, mol k₂cro₄, g k₂cro₄/mol
Step1: Dissociate $K_2CrO_4$ into ions
$K_2CrO_4
ightarrow 2K^+ + CrO_4^{2-}$
Step2: Set up mole ratio factor
Mole ratio: $\frac{2\ \text{mol } K^+}{1\ \text{mol } K_2CrO_4}$
Step3: Calculate $K^+$ molarity
$\frac{0.122\ \text{mol } K_2CrO_4}{\text{L}} \times \frac{2\ \text{mol } K^+}{1\ \text{mol } K_2CrO_4} = 0.244\ \text{mol } K^+/\text{L}$
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
0.244 M $K^+$