QUESTION IMAGE
Question
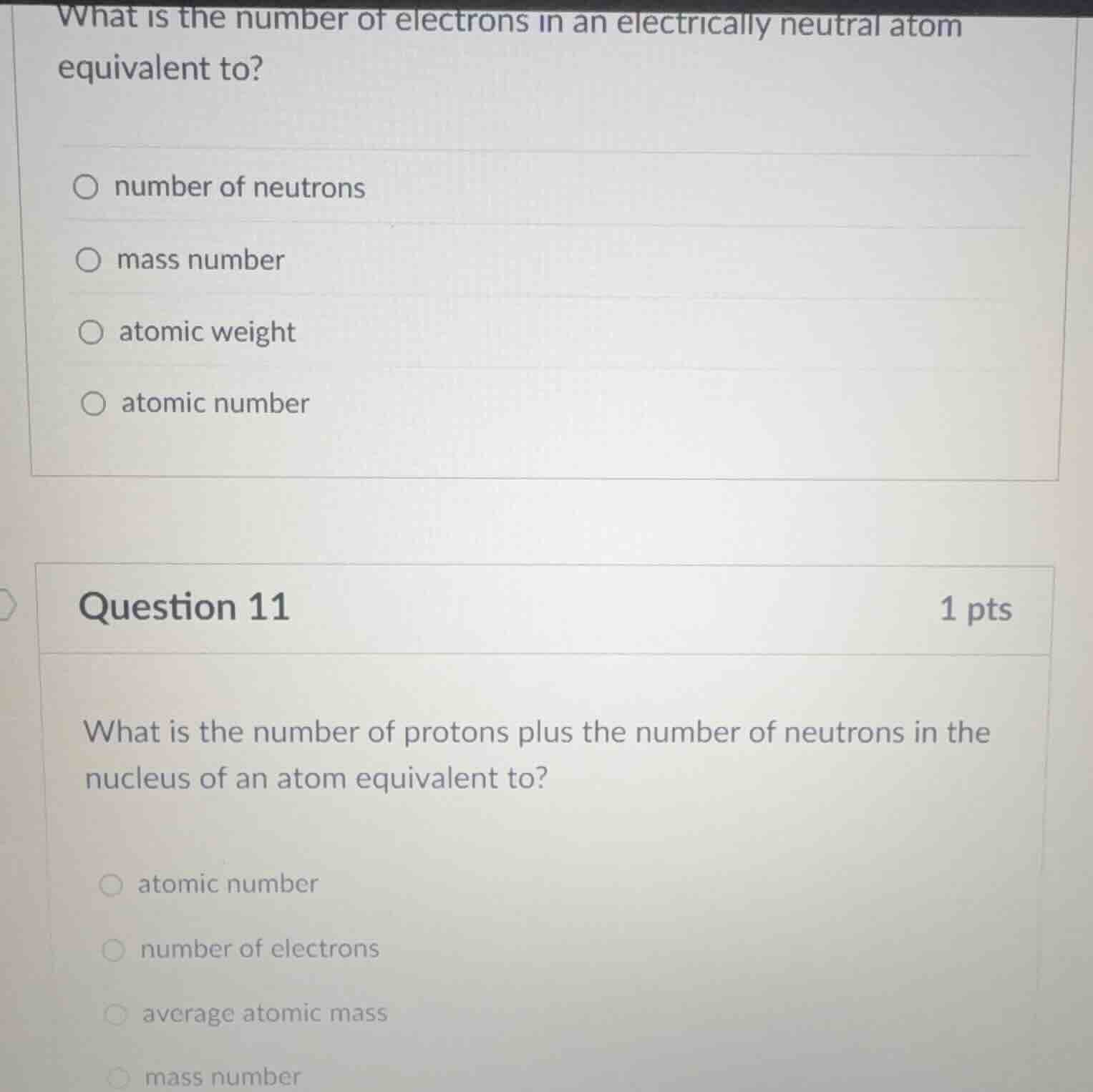
what is the number of electrons in an electrically neutral atom equivalent to?
number of neutrons
mass number
atomic weight
atomic number
question 11 1 pts
what is the number of protons plus the number of neutrons in the nucleus of an atom equivalent to?
atomic number
number of electrons
average atomic mass
mass number
First Question (Electrons in Neutral Atom)
In an electrically neutral atom, the number of electrons (negative charge) equals the number of protons (positive charge). The atomic number is defined as the number of protons in an atom. So, for a neutral atom, electrons = protons = atomic number. Neutrons are neutral, mass number is protons + neutrons, and atomic weight is an average mass.
The mass number of an atom is defined as the sum of the number of protons and neutrons in its nucleus. Atomic number is protons, electrons equal protons in neutral atoms, and average atomic mass is a weighted average of isotopes' masses.
Snap & solve any problem in the app
Get step-by-step solutions on Sovi AI
Photo-based solutions with guided steps
Explore more problems and detailed explanations
D. atomic number (assuming options are labeled A - D as: A. number of neutrons, B. mass number, C. atomic weight, D. atomic number)